-
PDF
- Split View
-
Views
-
Cite
Cite
György Széles, Zoltán Vokó, Tibor Jenei, László Kardos, Zsuzsa Pocsai, András Bajtay, Elemér Papp, Gabriella Pásti, Zsigmond Kósa, Ilona Molnár, Katalin Lun, Róza Ádány, A preliminary evaluation of a health monitoring programme in Hungary, European Journal of Public Health, Volume 15, Issue 1, February 2005, Pages 26–32, https://doi.org/10.1093/eurpub/cki107
Close - Share Icon Share
Abstract
Background: In 1998 a joint initiative of the Hungarian School of Public Health and the National Public Health Service created a network of sentinel stations based in primary care facilities in four Hungarian counties. The aim was to establish a system that will provide valid data on morbidity of selected diseases in Hungary. Methods: Based on standardized protocols, the participating centres have continuously reported data on the prevalence of cardiovascular diseases, diabetes mellitus, liver cirrhosis, and some malignant diseases, as well as supplying denominator data. The four counties represent both eastern and western parts of Hungary, reflecting the known geographical disparities in health. Each county office enrolled general practitioners maintaining representation in terms of both geography and distribution of settlement size. Results: A total of 73 general practitioners agreed to participate, providing care for 15.6% (138,088 people) of the population in the counties. The population registered with the practices were representative in terms of age and sex of both the participating counties and the entire country. The prevalence of hypertension, diabetes mellitus and liver cirrhosis is high in each county but varies considerably, with higher levels in the western counties, especially among older age groups of both sexes. Conclusions: The establishment of sentinel stations to collect morbidity data is feasible and sustainable in Hungarian primary care. The data that have been generated provide a valid and comprehensive picture of important aspects of the Hungarian population's health, with important implications for health policy and health service planning. In regions where low prevalence rates of diseases and high mortality rates simultaneously exist special attention is required to explore the background of this caveat.
Abstract
Key points
Till the end of 1998 no program operated in Hungary engaged with non-communicable disease morbidity data collection, except some hospital-based registries, which failed to produce reliable information.
The establishment of sentinel stations to collect morbidity data is feasible and sustainable in Hungarian primary care, the valid morbidity data can be built into the decision making process in health service planning.
Regular training, quality control and feedback are important contributors to the success of the program.
The prevalence of hypertension, diabetes mellitus and liver cirrhosis is high in each county but varies considerably, with higher levels in the western counties, especially among older age groups of both sexes.
More research needed to determine the possible contribution of unknown morbidity and health service utilisation to the different prevalence values in the two parts of Hungary.
As judged by levels of mortality, the health status of the Hungarian population compares poorly, not only in relation to Western Europe but even to its Central and Eastern European neighbours.1–5 However, mortality is an incomplete assessment of population health. Death is only the final stage in an often lengthy period of ill-health.6,7 Valid data on morbidity have many uses. They can inform the process of determining priorities, developing health policy, and evaluating effectiveness. There are now examples of successful systems based in primary care from many parts of the world.8–15 Ensuring the validity of morbidity data collected in primary care, as in registries based in secondary care, depends on motivating participants. At this time, with the sentinel station method, it is possible to collect data from a representative sample instead of the entire population, since general practitioners have information on the denominator and so can calculate the prevalence and incidence.
Essentially, there are two models for collecting morbidity data in primary care. One is based on episodes of care, recording data on all doctor–patient interactions, gathering information on consultation rates and patterns of clinical management; the other focuses on specific disorders, using a limited number of standardized case definitions and attempting to assess the burden of disease attributable to those disorders in the population in question. The first model is exemplified by the English General Practice Research Database Programme,11,16 and the use of ICPC codes,12 while the second one is illustrated by the Morbidity Sentinel Stations Programme that is now operational in several European countries.13–15 Both these approaches have shown how primary care records are a potentially rich source of morbidity data.17
Previous attempts to obtain morbidity data from general practitioners in Hungary were unsuccessful, to a considerable extent because they were seen as too labour intensive,18 a problem exacerbated by the many new and competing demands imposed by new funding and regulatory systems in the 1990s. Thus general practitioners already must report to the National Health Insurance Fund and the National Public Health and Medical Officer Service monthly. Unfortunately the existing data do not support epidemiological analyses. As a result they tend to be averse to collecting even more data and also expect to be compensated for their work.
However, since the previous unsuccessful introduction of a morbidity registration programme in primary care there has been a strong political commitment on behalf of decision–makers in the health sector in Hungary to assess more accurately the burden of disease in the population. In order to achieve this goal a system of sentinel stations was established. The objectives of our programme were to implement and evaluate a methodology that would collect valid, complete, continuous, and representative data on selected diseases of major public health importance. A subsequent goal was to ensure sustainability of the system and to build sufficient analytic capacity to be able to present the results in an appropriate format to health professionals and key decision-makers.
This paper aims to demonstrate the experience of establishing a primary care based Sentinel Station Programme, as well as describe the prevalence of three chronic diseases with high public health importance in Hungary: hypertension, diabetes mellitus and liver cirrhosis.
Methods
By 1998 the School of Public Health in the University of Debrecen and the National Public Health and Medical Officer Service had established computerized community laboratories in four county public health offices. The four counties (Hajdú-Bihar, Szabolcs-Szatmár-Bereg, Győr-Moson-Sopron, and Zala) were chosen to represent the eastern and western parts of Hungary (figure 1), reflecting well-recognized differences in measures of health, as well as in other factors such as economic activity. These laboratories formed the foci of the sentinel surveillance system.
Staff in the county offices were asked to recruit a sample of general practitioners in their county that was representative both geographically and in terms of settlement size included.
The major criteria for being selected were the possession of computer facilities, and commitment to participate in a continuous data collection programme. Each general practice had facilities for computerized data entry. Several issues were considered in designing the system. Recognizing the already high administrative load of general practitioners, the method of data collection should minimize their additional workload once the initial phase of assessing prevalence data was complete. The data should be standardized and the system should be logistically feasible, implying sampling rather than seeking to provide complete population coverage, and with a focus on a discrete list of conditions that were important in public health terms in Hungary. Data collection began in October 1998.
Study population
The study population includes all those registered with the participating practices. As the proportion of the population registered at general practitioners in Hungary is virtually 100%, it enables us to provide an accurate population denominator. Furthermore, inhabitants are registered to a single practitioner only, which eliminates the biasing effect of double counting. The enrolled general practices cover 138088 people (15.6% of the population in the counties concerned), of whom 47.9% were male and 52.1% female. The size of practices involved in the programme varies over a wide range (between 635 and 3195 inhabitants), the fact that there is a difference in the representation of participating practices and that of the population for which they provide services suggests under-representation of practices of small size. This is a consequence of the selection criteria applied by the staff of county offices who were asked to recruit a sample of general practitioners to represent the distribution of practices geographically and in terms of settlement size and not in terms of practice size.
Data collection and case definition
The programme collected data on the following diseases: hypertension, diabetes mellitus, liver cirrhosis, ischaemic heart disease (except myocardial infarction), acute myocardial infarction, stroke, malignant neoplasm of trachea, bronchi and lung, malignant neoplasm of colon and rectum, malignant neoplasm of breast, malignant neoplasm of cervix and malignant neoplasm of prostate. Standardized diagnostic criteria were developed, based on existing definitions. However, in many cases, especially cancer, diagnosis is likely to be made not in general practice but in hospital or specialist outpatient clinics. In these cases it was not possible to apply the standardized criteria and it was necessary for the general practitioner to use the diagnosis provided. Hypertension is defined as a systolic blood pressure of 140 mmHg or greater and/or a diastolic blood pressure of 90 mmHg or greater measured according to the WHO blood pressure measurement guidelines.19 The diagnosis of liver cirrhosis is based on a combination of symptoms and positive laboratory findings with at least two of the following symptoms: spider naevus, ascites, palmar and plantar erythema, jaundice, enlarged, firm liver with rounded or nodular edge, and at least one positive laboratory results: increased level of serum bilirubin, aspartate aminotransferase, alanine aminotransferase, gamma-glutamyl-transpeptidase, alkaline-phosphatase, decreased serum albumin, and prolonged prothrombin time. Finally, diagnosis of diabetes mellitus requires fasting plasma glucose level equal to or more than 7.8 mmol/l more than once and/or postprandial plasma glucose level equal to or more than 11.1 mmol/l, with other relevant characteristics of patients used to define the types of diabetes. The results of the first year quality control showed that separation of type I and II diabetes was not feasible as the diagnostic criteria varied greatly between general practitioners. After prevalence data analysis, we found that the county age–specific prevalence of type I diabetes was increasing with age in all counties, which contradicts its diagnostic criteria. Therefore we have monitored diabetes mellitus as such regardless of the classification. On recruitment, participating general practitioners provided data on patients who were known to have any of the relevant conditions as well as the age and gender breakdown of the practice population. Data on the composition of practice populations has been updated each January.
Each month, notifications of incident cases of any of the conditions included in the scheme is forwarded to the relevant county offices of the National Public Health and Medical Officer Service, where they are coded, using a taxonomy based on the Tenth Revision of the International Classification of Diseases, and data entry and primary analysis take place. Data are then forwarded quarterly to the School of Public Health. There might be some delay in incident data reporting but it has no major affect on the analysis as it is done only yearly. Incomplete reporting of new cases occurring in the practices is thought to be minimized by targeted and regular quality assurance checks involving each practice once every year. Furthermore this work is backed up by regular training. Initial experience indicates that about 10 new cases of the group of selected diseases are occurring in a typical practice on average each month.
The data supplied to the community laboratory includes the diagnosis (coded according to the International Classification of Diseases) and where and how the diagnosis was made (general practitioner, outpatient clinic, hospital, or autopsy). In addition, transfers in and out (whether on death or otherwise) of the practice are recorded. General practitioners are identified by a code used by the National Public Health and Medical Officer Service. Patients have a unique registration number given by the general practitioner at registration and that can only be decoded by the general practitioner, so preserving anonymity; on the other hand, data on multiple episodes can be linked.
Statistical analysis
Baseline age-sex specific prevalence by county was estimated after the initial round of data collection. Using the method of indirect standardisation, age/sex standardised prevalence ratios were calculated for the selected diseases and results were used for targeted quality assurance checks.
Data quality
This system depends on quality assurance at all levels. General practitioners undergo an initial one-day training course and receive regular feedback on both their data quality and the emerging results. They are also invited to six-monthly workshops with participating staff from the county office. Issues arising from these workshops are addressed in information letters sent to all participating general practitioners. Staff from the county office also visit each practice at least once a year to check systems of data collection. This involves a formal validity check in which data on 10 patients are checked. Five patients are chosen randomly from the county database (those patients who had been reported within the programme as having any of the selected diseases) and the other five selected by chance from the general practitioner's computer database, which includes information about all inhabitants registered at that time. For a given practice the proportion of files checked depends upon the actual number of inhabitants registered in that practice. Data entry also involves several automated checks, identifying values outside acceptable ranges and flagging duplicate entries (diagnoses can only be recorded once for each individual except in the case of stroke or myocardial infarction), although these also elicit queries to confirm reoccurrence. Finally, checks are triggered during analysis when practices are found to have reported standardized rates outside the range of +/- 50% of the expected value and/or the difference between expected and observed cases is ≥5 and/or the value for the standardized prevalence ratio is missing, or where an unexpected number of diagnoses originating from a particular setting emerges.
For most of the diseases in our study the diagnosis was made by specialists in outpatient clinics and hospitals, therefore we had to rely on the diagnostic algorithms applied in standard medical practice. At this stage of the programme the participating practitioners do not assess the validity of the diagnoses made by specialists.
Results
Eighty general practitioners selected by the county offices were invited to participate and 73 (7.9% of all practices in the four participating counties) agreed. The population was representative in terms of age and sex of both the participating counties and the overall Hungarian population (table 1).
After the initial prevalence analysis, targeted quality control visits were carried out in those practices where incorrect data submission was suspected. In those participating practices where targeted field checks were not necessary, regular quality control visits were carried out.
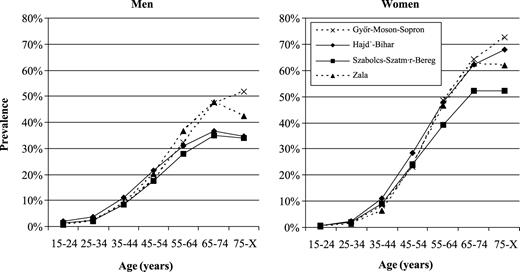
A consistent pattern emerges in which the prevalence of most diseases is highest in the two western counties (Győr-Moson-Sopron and Zala). The disjunction of the prevalence figures can be seen from the age group 55–64 years, which results in the greatest variation in prevalence figures at older ages in both sexes.
The prevalence of hypertension increases steeply with age showing higher prevalence in females from the age 45 years upwards. The prevalence of hypertension was highest in the two western counties (around 50%) among 65–74 year old men, whereas it was less than 40% among a similar aged male population in the two eastern counties (figure 2). Among women in this age group the prevalence of hypertension exceeded 60% in the two western counties and in one of the eastern counties (Hajdú-Bihar), while in the other eastern county half of the women in this age group suffered from hypertension.
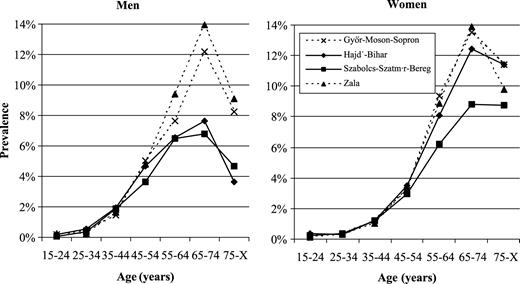
The prevalence of diabetes also increases with age, although it declines beyond the age group 65–74, presumably reflecting a higher premature mortality among those with diabetics than the general population. The prevalence of diabetes mellitus was found to be significantly higher in the western counties compared to the eastern counties among men aged 45 years or older (figure 3). The biggest difference was observed in the age group 65–74, where the prevalence of diabetes mellitus was above 10% in the western counties (12.2% and 13.9% respectively) but well below 10% in the eastern counties (7.6% and 6.8% respectively). Among women, as with the prevalence of hypertension, the same eastern county (Hajdú–Bihar) approximated the western counties, all three having higher than 10% prevalence in the age group 65–74 years. The prevalence of diabetes mellitus in men living in the eastern part of Hungary was significantly lower compared to women in the east, in the older age groups.
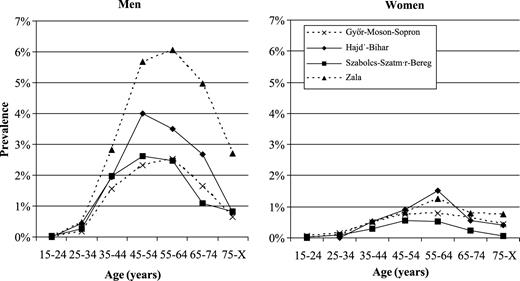
There was a wide variation in the prevalence of liver cirrhosis between participating counties among both males and females (figure 4). The prevalence was consistently higher among males in all counties. The prevalence of liver cirrhosis was found to be 1.5–2.5 times higher in Zala county (one of the western counties) compared to the other three counties among middle aged men (35–64 years) and the difference was even higher in the oldest age group (65+years). The difference in the prevalence of liver cirrhosis among women was greatest in the age group 55–64 years, with the highest result observed in one eastern county (Hajdú-Bihar: 1.5%) and the lowest figure was seen in the other eastern county (Szabolcs-Szatmár–Bereg: 0.5%). The consistent east–west gap observed in the case of hypertension and diabetes mellitus was not seen with liver cirrhosis.
Discussion
As Fleming has noted, morbidity data collected in general practices are of great value, as general practitioners are gatekeepers to the rest of the health care system.11 Drawing on extensive experience, he emphasizes that morbidity data should be based on individuals rather than consultations, as while the latter may provide information on workload, they have limited epidemiological value.
The programme described here offers the possibility to collect large volumes of morbidity data from representative population samples in different geographical regions in Hungary. Representativeness of practices and patients is an important issue in judging the external validity of the results. Although some literature reports that voluntary registries do not violate the validity of study results,20 there is a strong argument that representativeness of the study sample has a serious effect on the generelizability of outcomes.8,21 In our morbidity programme, participating general practitioners produce a sample that is representative of the age and sex distribution of the participating counties.
The internal validity of the programme mainly depends upon the data registration/collection, coding system and the sources of diagnosis. One of the key issues is minimizing the workload of general practitioners caused by additional data recording. This can be achieved if the registration system is part of the general patient registration system, linked to an appropriate computer package. In our programme only the process of collecting data on prevalence created a major time burden on general practitioners, this being confined to the beginning of the study. Subsequent monthly incidence reporting requires significantly less time to report, and results in about 3 hours per month additional workload.
As one of our main objectives is to ensure the accuracy of diagnosis, we use the International Classification of Diseases (ICD) coding system knowing that it might not satisfy the requirements of a highly developed general practice-based information system such as the International Classification of Primary Care (ICPC).22 In fact, Hungary considered introducing an ICPC-based registration system in primary health care between 1996 and 1997 but the results of the programme are questionable, with numerous weaknesses concerning the internal validity of the data, such as the lack of well-defined standardized diagnostic criteria. There is little information about how the Hungarian adaptation of the ICPC system was undertaken18 but that programme sought to measure every episode of any disease in general practice. Furthermore, it is not clear how the system handled the immigrated cases and deaths.
In our study, where diagnosis was mostly made by specialists in outpatient clinics and hospitals, we had to rely on the diagnostic algorithms applied in standard medical practice. We believe that this is unlikely to cause major bias in cases of diabetes mellitus, hypertension, malignant diseases, myocardial infraction and stroke because the diagnoses are made according to generally accepted principles in Hungarian medical practice. However, we accept that this requires further validation. In the case of ischaemic heart disease and cirrhosis of the liver, medical practice is not uniform and bias seems likely.
During preparation of the study, alcoholic liver disease arose as an important issue. However, because of diagnostic variations in hospital practice, we chose cirrhosis of the liver, which has more exact diagnostic criteria. Yet even here, of necessity, the criteria adopted reflect the needs of the system, based on investigations available to the general practitioner rather than, for example, using histological criteria applied to liver biopsy, which might be considered the gold standard.
The prevalence of hypertension, diabetes mellitus and liver cirrhosis is high in all the counties involved. In most cases we have found geographical variations, showing different values in the eastern and western parts of Hungary, with higher values in the western counties. The observed differences may be real variation in prevalence, but survival time and proportion of cases identified by the health services may also be important. High premature mortality could partially explain the lower prevalence in the eastern part of the country, especially in Szabolcs-Szatmár-Bereg where a recent study showed one of the highest risks of early death.5
Differences in the quality of data provided by general practitioners in different counties can also explain the differences observed in the prevalence of the selected diseases between the counties, which was found to be negligible based upon the quality control visits. Furthermore, comparison of the prevalence data with the cause-specific mortality figures of these counties (with the worst results in the eastern counties) clearly suggests a significant role for the unknown morbidity among people living in the eastern part of Hungary. This issue will be resolved once more incidence data are available but exploration of these potential causes is crucial to explain the differences between different regions and to plan affective preventive measures. Finally, the difference in the prevalence figures might be a reflection of the different distribution of unmeasured confounding factors such as social, economical and health behavioural determinants.
The higher increasing hypertension prevalence among females might be explained by the very unfavourable health conditions and subsequent higher premature mortality of Hungarian men, which is a well-known fact. This assumption is further strengthened by the more sharply dropping prevalence figures in the other two conditions among males after 55 years of age.
The European Commission's recent report on Health Monitoring in Sentinel Practice Networks project reports the prevalence of diabetes mellitus in the year 2000 in six different countries.23 The only age group of which prevalence date are comparable with our data is 65–74, where the prevalence of diabetes mellitus among men living in the two western counties in Hungary (Győr-Moson-Sopron county 12.2% and Zala county 13.9%) was higher than the two countries (Belgium 11.2% and Spain (País Vasco) 11%) with the highest period prevalence in this age group (table 2). In the two eastern counties participating in the Hungarian General Practitioners’ Morbidity Sentinel Stations Programme, in men aged 65–74 years the prevalence of diabetes mellitus did not show major differences from those countries participating in the Sentinel Practice Network. Among females in this age group, the prevalence of diabetes showed substantially higher values in the two western counties (Győr-Moson-Sopron and Zala) and one of the eastern counties (Hajdú-Bihar) in Hungary (table 3).
Our experience so far indicates that a sentinel surveillance system based in general practice is feasible in Hungary. Given the lack of familiarity with this concept, regular training, quality control and feedback have been emphasized and have contributed much to the success of the first phase of the project. In the future we plan to extend the scope of the programme and to integrate the data with the process of health service planning. This programme may serve as a demonstration project for Hungary and provides a sound basis for postgraduate public health training and longitudinal research in general practice.
We would like to express our gratitude for the continuous data collection effort of general practitioners and also for the tireless work provided by the public health county office workers. The authors are also grateful to Professor Martin McKee for his valuable comments on the earlier versions of this manuscript.
This project was funded partially by the Hungarian Ministry of Health and a grant from the Scientific Health Council, ETT 09/588.
The participating counties in the General Practitioners’ Morbidity Sentinel Stations Programme in Hungary
Prevalence of hypertension in four counties in Hungary by gender and age in 1998
Prevalence of diabetes mellitus in four counties in Hungary by gender and age in 1998
Prevalence of liver cirrhosis in four counties in Hungary by gender and age in 1998
Age and sex distribution of practices involved in the General Practitioners’ Morbidity Sentinel Stations Programme and of the four counties and the Hungarian populations aged 15 and older
| All four counties . | Percentage of sample practices (n=127 411) . | Percentage of counties population (n=1 490 652) . | Percentage of Hungarian population (n=8 347 163) . |
|---|---|---|---|
| Males: . | (n=60 592) . | (n=713 500) . | (n=3 924 145) . |
| 15–24 | 17.3 | 21.2 | 20.4 |
| 25–34 | 18.6 | 18.2 | 17.8 |
| 35–44 | 18.8 | 19.0 | 18.0 |
| 45–54 | 17.6 | 17.0 | 17.4 |
| 55–64 | 12.3 | 11.4 | 12.3 |
| 65–74 | 9.9 | 8.9 | 9.2 |
| 75–X | 5.4 | 4.3 | 4.7 |
| Females: | (n=66 819) | (n=777 152) | (n=4 423 018) |
| 15–24 | 15.2 | 18.4 | 17.3 |
| 25–34 | 16.4 | 15.5 | 15.2 |
| 35–44 | 16.8 | 17.0 | 16.2 |
| 45–54 | 16.4 | 16.1 | 16.7 |
| 55–64 | 13.6 | 13.2 | 13.8 |
| 65–74 | 12.7 | 12.1 | 12.3 |
| 75–X | 8.9 | 7.7 | 8.9 |
| All four counties . | Percentage of sample practices (n=127 411) . | Percentage of counties population (n=1 490 652) . | Percentage of Hungarian population (n=8 347 163) . |
|---|---|---|---|
| Males: . | (n=60 592) . | (n=713 500) . | (n=3 924 145) . |
| 15–24 | 17.3 | 21.2 | 20.4 |
| 25–34 | 18.6 | 18.2 | 17.8 |
| 35–44 | 18.8 | 19.0 | 18.0 |
| 45–54 | 17.6 | 17.0 | 17.4 |
| 55–64 | 12.3 | 11.4 | 12.3 |
| 65–74 | 9.9 | 8.9 | 9.2 |
| 75–X | 5.4 | 4.3 | 4.7 |
| Females: | (n=66 819) | (n=777 152) | (n=4 423 018) |
| 15–24 | 15.2 | 18.4 | 17.3 |
| 25–34 | 16.4 | 15.5 | 15.2 |
| 35–44 | 16.8 | 17.0 | 16.2 |
| 45–54 | 16.4 | 16.1 | 16.7 |
| 55–64 | 13.6 | 13.2 | 13.8 |
| 65–74 | 12.7 | 12.1 | 12.3 |
| 75–X | 8.9 | 7.7 | 8.9 |
Age and sex distribution of practices involved in the General Practitioners’ Morbidity Sentinel Stations Programme and of the four counties and the Hungarian populations aged 15 and older
| All four counties . | Percentage of sample practices (n=127 411) . | Percentage of counties population (n=1 490 652) . | Percentage of Hungarian population (n=8 347 163) . |
|---|---|---|---|
| Males: . | (n=60 592) . | (n=713 500) . | (n=3 924 145) . |
| 15–24 | 17.3 | 21.2 | 20.4 |
| 25–34 | 18.6 | 18.2 | 17.8 |
| 35–44 | 18.8 | 19.0 | 18.0 |
| 45–54 | 17.6 | 17.0 | 17.4 |
| 55–64 | 12.3 | 11.4 | 12.3 |
| 65–74 | 9.9 | 8.9 | 9.2 |
| 75–X | 5.4 | 4.3 | 4.7 |
| Females: | (n=66 819) | (n=777 152) | (n=4 423 018) |
| 15–24 | 15.2 | 18.4 | 17.3 |
| 25–34 | 16.4 | 15.5 | 15.2 |
| 35–44 | 16.8 | 17.0 | 16.2 |
| 45–54 | 16.4 | 16.1 | 16.7 |
| 55–64 | 13.6 | 13.2 | 13.8 |
| 65–74 | 12.7 | 12.1 | 12.3 |
| 75–X | 8.9 | 7.7 | 8.9 |
| All four counties . | Percentage of sample practices (n=127 411) . | Percentage of counties population (n=1 490 652) . | Percentage of Hungarian population (n=8 347 163) . |
|---|---|---|---|
| Males: . | (n=60 592) . | (n=713 500) . | (n=3 924 145) . |
| 15–24 | 17.3 | 21.2 | 20.4 |
| 25–34 | 18.6 | 18.2 | 17.8 |
| 35–44 | 18.8 | 19.0 | 18.0 |
| 45–54 | 17.6 | 17.0 | 17.4 |
| 55–64 | 12.3 | 11.4 | 12.3 |
| 65–74 | 9.9 | 8.9 | 9.2 |
| 75–X | 5.4 | 4.3 | 4.7 |
| Females: | (n=66 819) | (n=777 152) | (n=4 423 018) |
| 15–24 | 15.2 | 18.4 | 17.3 |
| 25–34 | 16.4 | 15.5 | 15.2 |
| 35–44 | 16.8 | 17.0 | 16.2 |
| 45–54 | 16.4 | 16.1 | 16.7 |
| 55–64 | 13.6 | 13.2 | 13.8 |
| 65–74 | 12.7 | 12.1 | 12.3 |
| 75–X | 8.9 | 7.7 | 8.9 |
Prevalence of diabetes mellitus in the Hungarian General Practitioners’ Morbidity Sentinel Stations Programme and the Health Monitoring Sentinel Practice Networks among males aged 65–74
| Hungarian counties . | Prevalence (%) . | . | Sentinel Practice Networks . | |
|---|---|---|---|---|
| . | 1998 . | 2000 . | . | |
| Győr-Moson-Sopron | 12.2 | 7.7 | England and Wales | |
| Zala | 13.9 | 11.2 | Belgium | |
| 8.0 | Portugal | |||
| Hajdú-Bihar | 7.6 | 7.1 | Spain (Castilla y Léon) | |
| 11.0 | Spain (País Vasco) | |||
| Szabolcs-Szatmár-Bereg | 6.8 | 5.9 | Spain (Comunitat Valenciana) | |
| 9.9 | France | |||
| Hungarian counties . | Prevalence (%) . | . | Sentinel Practice Networks . | |
|---|---|---|---|---|
| . | 1998 . | 2000 . | . | |
| Győr-Moson-Sopron | 12.2 | 7.7 | England and Wales | |
| Zala | 13.9 | 11.2 | Belgium | |
| 8.0 | Portugal | |||
| Hajdú-Bihar | 7.6 | 7.1 | Spain (Castilla y Léon) | |
| 11.0 | Spain (País Vasco) | |||
| Szabolcs-Szatmár-Bereg | 6.8 | 5.9 | Spain (Comunitat Valenciana) | |
| 9.9 | France | |||
Prevalence of diabetes mellitus in the Hungarian General Practitioners’ Morbidity Sentinel Stations Programme and the Health Monitoring Sentinel Practice Networks among males aged 65–74
| Hungarian counties . | Prevalence (%) . | . | Sentinel Practice Networks . | |
|---|---|---|---|---|
| . | 1998 . | 2000 . | . | |
| Győr-Moson-Sopron | 12.2 | 7.7 | England and Wales | |
| Zala | 13.9 | 11.2 | Belgium | |
| 8.0 | Portugal | |||
| Hajdú-Bihar | 7.6 | 7.1 | Spain (Castilla y Léon) | |
| 11.0 | Spain (País Vasco) | |||
| Szabolcs-Szatmár-Bereg | 6.8 | 5.9 | Spain (Comunitat Valenciana) | |
| 9.9 | France | |||
| Hungarian counties . | Prevalence (%) . | . | Sentinel Practice Networks . | |
|---|---|---|---|---|
| . | 1998 . | 2000 . | . | |
| Győr-Moson-Sopron | 12.2 | 7.7 | England and Wales | |
| Zala | 13.9 | 11.2 | Belgium | |
| 8.0 | Portugal | |||
| Hajdú-Bihar | 7.6 | 7.1 | Spain (Castilla y Léon) | |
| 11.0 | Spain (País Vasco) | |||
| Szabolcs-Szatmár-Bereg | 6.8 | 5.9 | Spain (Comunitat Valenciana) | |
| 9.9 | France | |||
Prevalence of diabetes mellitus in the Hungarian General Practitioners’ Morbidity Sentinel Stations Programme and the Health Monitoring Sentinel Practice Networks among females aged 65–74
| Hungarian counties . | Prevalence (%) . | . | Sentinel Practice Networks . | |
|---|---|---|---|---|
| . | 1998 . | 2000 . | . | |
| Győr-Moson-Sopron | 13.5 | 5.6 | England and Wales | |
| Zala | 13.9 | 9.6 | Belgium | |
| 7.1 | Portugal | |||
| Hajdú-Bihar | 12.5 | 9.5 | Spain (Castilla y Léon) | |
| 11.0 | Spain (País Vasco) | |||
| Szabolcs-Szatmár-Bereg | 8.8 | 8.0 | Spain (Comunitat Valenciana) | |
| 6.7 | France | |||
| Hungarian counties . | Prevalence (%) . | . | Sentinel Practice Networks . | |
|---|---|---|---|---|
| . | 1998 . | 2000 . | . | |
| Győr-Moson-Sopron | 13.5 | 5.6 | England and Wales | |
| Zala | 13.9 | 9.6 | Belgium | |
| 7.1 | Portugal | |||
| Hajdú-Bihar | 12.5 | 9.5 | Spain (Castilla y Léon) | |
| 11.0 | Spain (País Vasco) | |||
| Szabolcs-Szatmár-Bereg | 8.8 | 8.0 | Spain (Comunitat Valenciana) | |
| 6.7 | France | |||
Prevalence of diabetes mellitus in the Hungarian General Practitioners’ Morbidity Sentinel Stations Programme and the Health Monitoring Sentinel Practice Networks among females aged 65–74
| Hungarian counties . | Prevalence (%) . | . | Sentinel Practice Networks . | |
|---|---|---|---|---|
| . | 1998 . | 2000 . | . | |
| Győr-Moson-Sopron | 13.5 | 5.6 | England and Wales | |
| Zala | 13.9 | 9.6 | Belgium | |
| 7.1 | Portugal | |||
| Hajdú-Bihar | 12.5 | 9.5 | Spain (Castilla y Léon) | |
| 11.0 | Spain (País Vasco) | |||
| Szabolcs-Szatmár-Bereg | 8.8 | 8.0 | Spain (Comunitat Valenciana) | |
| 6.7 | France | |||
| Hungarian counties . | Prevalence (%) . | . | Sentinel Practice Networks . | |
|---|---|---|---|---|
| . | 1998 . | 2000 . | . | |
| Győr-Moson-Sopron | 13.5 | 5.6 | England and Wales | |
| Zala | 13.9 | 9.6 | Belgium | |
| 7.1 | Portugal | |||
| Hajdú-Bihar | 12.5 | 9.5 | Spain (Castilla y Léon) | |
| 11.0 | Spain (País Vasco) | |||
| Szabolcs-Szatmár-Bereg | 8.8 | 8.0 | Spain (Comunitat Valenciana) | |
| 6.7 | France | |||
References
Boján F, Hajdú P, Belicza E. Regional differences in avoidable mortality in Europe. In: Normand CEM, Vaughan JP, editors. Europe without frontiers–the implications for health. Chichester: John Wiley and Sons,
Hajdú P, McKee M, Boján F. Changes in premature mortality differentials by marital status in Hungary and in England and Wales.
Varvasovszky Z, Bain C, McKee M. Alcohol related mortality in Poland and Hungary: differences and similarities.
European Observatory on Health Care Systems. Health care systems in transition: Hungary. Copenhagen: European Observatory on Health Care Systems,
Vargáné Hajdú, P., Ádány, R. A halálozás területi különbségei Magyarországon, 1994-1996 (Mortality in Hungary at regional levels, 1994-1996.) [In Hungarian]. Népegészségügy 2000;81:4-26.
Declich S, Carter AO. Public health surveillance: historical origins, methods and evaluation.
O'Toole B, Driver B, Britt H, Bridges-Webb C. Using general practitioners to measure community morbidity.
Pearson N, O Brien J, Thomas H, Ewings P, Gallier L, Bussey A. Collecting morbidity data in general practice: the Somerset morbidity project.
Fleming DM. The measurement of morbidity in general practice.
Brage S, Bentsen BG, Bjerkedal T, Nygard JF, Tellnes G. ICPC as a standard classification in Norway.
Chauvin P, Valleron A. Participation of French general practitioners in public health surveillance: a multidisciplinary approach.
Milne RM, Taylor MW, Taylor RJ. Audit of populations in general practice: the creation of a national resource for the study of morbidity in Scottish general practice.
de Grauw WJC, van den Hoogen HJM, van de Lisdonk EH, van Gerwen WHEM, van Weel C. Control group characteristics and study outcomes: empirical data from a study on mortality of patients with type 2 diabetes mellitus in Dutch general practice.
Országos Háziorvosi Intézet. Morbiditás regisztrációs program végső jelentés (Final report of the morbidity registration programme.) [In Hungarian]. Budapest: Országos Háziorvosi Intézet, 1997.
WHO Expert Committee. Hypertension control. WHO Tech. Rep. Ser. no. 862. Geneva: World Health Organization,
Cockburn J, Campbell E, Gordon JJ, Sanson-Fisher RW. Response bias in a study of general practice.
Fleming D. An epidemiological method applied to practices to measure the representativeness of their prescribing characteristics.
Hofmans-Okkes IM, Lamberts H. The International Classification of Primary Care (ICPC): new applications in research and computer-based patient records in family practice.




Comments