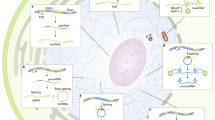
Key Points
-
Long non-coding RNAs (lncRNAs), which constitute a substantial fraction of mammalian transcriptomes, are new, enigmatic players in the complex transcriptional milieu.
-
RNA is a versatile molecule that is well suited for a myriad of functions. This versatility stems from adeptness of RNA at sequence-specific nucleic acid recognition, its ability to fold into intricate three-dimensional structures, as well as its dynamic and malleable nature.
-
lncRNAs directly and indirectly regulate transcription. Specifically, lncRNAs both positively and negatively influence transcription by modulating chromatin, acting as enhancers, regulating transcription factor function and organizing nuclear domains.
-
lncRNAs regulate mRNA processing by modulating pre-mRNA splicing and have the potential to direct mRNA editing. Additionally, lncRNAs modulate other post-transcriptional events such as translation, mRNA stability and miRNA-mediated repression.
-
lncRNAs can act as scaffolds to organize multiprotein complexes and subnuclear domains.
-
With lncRNAs now found in exosomes, lncRNAs have the potential to function as signalling molecules. In addition, lncRNAs can increase genetic diversity by determining the sites of recombination.
Abstract
The increased application of transcriptome-wide profiling approaches has led to an explosion in the number of documented long non-coding RNAs (lncRNAs). While these new and enigmatic players in the complex transcriptional milieu are encoded by a significant proportion of the genome, their functions are mostly unknown. Early discoveries support a paradigm in which lncRNAs regulate transcription via chromatin modulation, but new functions are steadily emerging. Given the biochemical versatility of RNA, lncRNAs may be used for various tasks, including post-transcriptional regulation, organization of protein complexes, cell-cell signalling and allosteric regulation of proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
The ENCODE Project Consortium. An integrated encyclopedia of DNA elements in the human genome. Nature 489, 57–74 (2012). Provides an overview of a series of papers released as part of the ENCODE project in which landmarks of biochemical function (regions of transcription, transcription factor association and histone modifications, among others) were attributed to 80% of the genome.
Carninci, P. et al. The transcriptional landscape of the mammalian genome. Science 309, 1559–1563 (2005).
Koch, F., Jourquin, F., Ferrier, P. & Andrau, J.-C. Genome-wide RNA polymerase II: not genes only! Trends Biochem. Sci. 33, 265–273 (2008).
Djebali, S. et al. Landscape of transcription in human cells. Nature 489, 101–108 (2012).
Taft, R. J., Pheasant, M. & Mattick, J. S. The relationship between non-protein-coding DNA and eukaryotic complexity. Bioessays 29, 288–299 (2007).
Berretta, J. & Morillon, A. Pervasive transcription constitutes a new level of eukaryotic genome regulation. EMBO Rep. 10, 973–982 (2009).
Mercer, T. R., Dinger, M. E. & Mattick, J. S. Long non-coding RNAs: insights into functions. Nature Rev. Genet. 10, 155–159 (2009).
Rinn, J. L. & Chang, H. Y. Genome regulation by long noncoding RNAs. Annu. Rev. Biochem. 81, 145–166 (2012).
Mercer, T. R. & Mattick, J. S. Structure and function of long noncoding RNAs in epigenetic regulation. Nature Struct. Mol. Biol. 20, 300–307 (2013).
Ponting, C. P. & Belgard, T. G. Transcribed dark matter: meaning or myth? Hum. Mol. Genet. 19, R162–R168 (2010).
Ponting, C. P., Oliver, P. L. & Reik, W. Evolution and functions of long noncoding RNAs. Cell 136, 629–641 (2009).
Wilusz, J. E., Sunwoo, H. & Spector, D. L. Long noncoding RNAs: functional surprises from the RNA world. Genes Dev. 23, 1494–1504 (2009).
Nagano, T. & Fraser, P. No-nonsense functions for long noncoding RNAs. Cell 145, 178–181 (2011).
Lee, J. T. Epigenetic regulation by long noncoding RNAs. Science 338, 1435–1439 (2012).
Kugel, J. F. & Goodrich, J. A. Non-coding RNAs: key regulators of mammalian transcription. Trends Biochem. Sci. 37, 144–151 (2012).
Robertson, M. P. & Joyce, G. F. The origins of the RNA world. Cold Spring Harb. Perspect. Biol. 4, pii: a003608. (2012).
Filipovska, A. & Rackham, O. Modular recognition of nucleic acids by PUF, TALE and PPR proteins. Mol. Biosyst 8, 699–708 (2012).
Stoltenburg, R., Reinemann, C. & Strehlitz, B. SELEX — a (r)evolutionary method to generate high-affinity nucleic acid ligands. Biomol. Eng. 24, 381–403 (2007).
Serganov, A. & Patel, D. J. Molecular recognition and function of riboswitches. Curr. Opin. Struct. Biol. 22, 279–286 (2012).
Pei, B. et al. The GENCODE pseudogene resource. Genome Biol. 13, R51 (2012).
Stoye, J. P. Studies of endogenous retroviruses reveal a continuing evolutionary saga. Nature Rev. Microbiol. 10, 395–406 (2012).
Khalil, A. M. et al. Many human large intergenic noncoding RNAs associate with chromatin-modifying complexes and affect gene expression. Proc. Natl Acad. Sci. USA 106, 11667–11672 (2009).
Zhao, J. et al. Genome-wide identification of polycomb-associated RNAs by RIP-seq. Mol. Cell 40, 939–953 (2010).
Tsai, M.-C. et al. Long noncoding RNA as modular scaffold of histone modification complexes. Science 329, 689–693 (2010). Illustrates an elegant example of a dominant theme in the lncRNA field whereby lncRNAs physically associate with histone-modifying complexes to regulate chromatin states. Importantly, shows that HOTAIR can also act as a scaffold to organize the concerted actions of two enzymatic activities.
Spitale, R. C., Tsai, M.-C. & Chang, H. Y. RNA templating the epigenome: long noncoding RNAs as molecular scaffolds. Epigenetics 6, 539–543 (2011).
Yap, K. L. et al. Molecular interplay of the noncoding RNA ANRIL and methylated histone H3 lysine 27 by polycomb CBX7 in transcriptional silencing of INK4a. Mol. Cell 38, 662–674 (2010).
Pandey, R. R. et al. Kcnq1ot1 antisense noncoding RNA mediates lineage-specific transcriptional silencing through chromatin-level regulation. Mol. Cell 32, 232–246 (2008).
Kotake, Y. et al. Long non-coding RNA ANRIL is required for the PRC2 recruitment to and silencing of p15INK4B tumor suppressor gene. Oncogene 30, 1956–1962 (2011).
Kornienko, A. E., Guenzl, P. M., Barlow, D. P. & Pauler, F. M. Gene regulation by the act of long non-coding RNA transcription. BMC Biol. 11, 59 (2013).
Latos, P. A. et al. Airn transcriptional overlap, but not its lncRNA products, induces imprinted Igf2r silencing. Science 338, 1469–1472 (2012). Provides a particularly compelling example of a mammalian lncRNA, AIRN , which represses target expression by transcriptional interference, as transcriptional overlap of the lncRNA with the target promoter rather than the lncRNA transcript itself is sufficient to interfere with Pol II recruitment.
Hongay, C. F., Grisafi, P. L., Galitski, T. & Fink, G. R. Antisense transcription controls cell fate in Saccharomyces cerevisiae. Cell 127, 735–745 (2006).
van Werven, F. J. et al. Transcription of two long noncoding RNAs mediates mating-type control of gametogenesis in budding yeast. Cell 150, 1170–1181 (2012).
Camblong, J., Iglesias, N., Fickentscher, C., Dieppois, G. & Stutz, F. Antisense RNA stabilization induces transcriptional gene silencing via histone deacetylation in S. cerevisiae. Cell 131, 706–717 (2007).
Carrozza, M. J. et al. Histone H3 methylation by Set2 directs deacetylation of coding regions by Rpd3S to suppress spurious intragenic transcription. Cell 123, 581–592 (2005).
Houseley, J., Rubbi, L., Grunstein, M., Tollervey, D. & Vogelauer, M. A ncRNA modulates histone modification and mRNA induction in the yeast GAL gene cluster. Mol. Cell 32, 685–695 (2008).
Schmitt, S., Prestel, M. & Paro, R. Intergenic transcription through a polycomb group response element counteracts silencing. Genes Dev. 19, 697–708 (2005).
Bumgarner, S. L. et al. Single-cell analysis reveals that noncoding RNAs contribute to clonal heterogeneity by modulating transcription factor recruitment. Mol. Cell 45, 470–482 (2012).
Hainer, S. J., Pruneski, J. A., Mitchell, R. D., Monteverde, R. M. & Martens, J. A. Intergenic transcription causes repression by directing nucleosome assembly. Genes Dev. 25, 29–40 (2011). Demonstrates a novel mode of SER3 gene repression by the yeast lncRNA SRG1 that involves the direction of nucleosome occupancy at the SER3 promoter.
Hirota, K. et al. Stepwise chromatin remodelling by a cascade of transcription initiation of non-coding RNAs. Nature 456, 130–134 (2008).
Lefevre, P., Witham, J., Lacroix, C. E., Cockerill, P. N. & Bonifer, C. The LPS-induced transcriptional upregulation of the chicken lysozyme locus involves CTCF eviction and noncoding RNA transcription. Mol. Cell 32, 129–139 (2008).
Bumgarner, S. L., Dowell, R. D., Grisafi, P., Gifford, D. K. & Fink, G. R. Toggle involving cis-interfering noncoding RNAs controls variegated gene expression in yeast. Proc. Natl Acad. Sci. USA 106, 18321–18326 (2009). Provides the first description of the pair of cis -interfering lncRNAs at the FLO11 locus in yeast, where a regulatory circuit toggles between two states of expression depending on the identity of the lncRNA expressed.
Flynn, R. A. & Chang, H. Y. Active chromatin and noncoding RNAs: an intimate relationship. Curr. Opin. Genet. Dev. 22, 172–178 (2012).
Ørom, U. A. et al. Long noncoding RNAs with enhancer-like function in human cells. Cell 143, 46–58 (2010). Describes a novel class of lncRNAs that, instead of repressing, activates target genes to function as RNA-dependent enhancers of gene expression.
Lai, F. et al. Activating RNAs associate with Mediator to enhance chromatin architecture and transcription. Nature 494, 497–501 (2013).
Kim, T.-K. et al. Widespread transcription at neuronal activity-regulated enhancers. Nature 465, 182–187 (2010).
Wang, D. et al. Reprogramming transcription by distinct classes of enhancers functionally defined by eRNA. Nature 474, 390–394 (2011).
Mariner, P. D. et al. Human Alu RNA is a modular transacting repressor of mRNA transcription during heat shock. Mol. Cell 29, 499–509 (2008).
Martianov, I., Ramadass, A., Serra Barros, A., Chow, N. & Akoulitchev, A. Repression of the human dihydrofolate reductase gene by a non-coding interfering transcript. Nature 445, 666–670 (2007).
Shamovsky, I., Ivannikov, M., Kandel, E. S., Gershon, D. & Nudler, E. RNA-mediated response to heat shock in mammalian cells. Nature 440, 556–560 (2006).
Kino, T., Hurt, D. E., Ichijo, T., Nader, N. & Chrousos, G. P. Noncoding RNA gas5 is a growth arrest- and starvation-associated repressor of the glucocorticoid receptor. Sci Signal 3, ra8 (2010).
Feng, J. et al. The Evf-2 noncoding RNA is transcribed from the Dlx-5/6 ultraconserved region and functions as a Dlx-2 transcriptional coactivator. Genes Dev. 20, 1470–1484 (2006).
Bond, A. M. et al. Balanced gene regulation by an embryonic brain ncRNA is critical for adult hippocampal GABA circuitry. Nature Neurosci. 12, 1020–1027 (2009).
Wang, X. et al. Induced ncRNAs allosterically modify RNA-binding proteins in cis to inhibit transcription. Nature 454, 126–130 (2008).
Willingham, A. T. et al. A strategy for probing the function of noncoding RNAs finds a repressor of NFAT. Science 309, 1570–1573 (2005). The first study to functionally screen 512 evolutionarily conserved putative lncRNAs. Identified the NRON lncRNA as a repressor of NFAT nuclear trafficking.
Yang, L. et al. ncRNA- and Pc2 methylation-dependent gene relocation between nuclear structures mediates gene activation programs. Cell 147, 773–788 (2011). Describes a particularly interesting paradigm whereby the post-translational modification status of a protein effector constitutes a switch in lncRNA-binding specificity and consequently determines the nuclear subdomain localization of target genes.
Geisler, S., Lojek, L., Khalil, A. M., Baker, K. E. & Coller, J. Decapping of long noncoding RNAs regulates inducible genes. Mol. Cell 45, 279–291 (2012).
Mattick, J. S. Non-coding RNAs: the architects of eukaryotic complexity. EMBO Rep. 2, 986–991 (2001).
Moore, M. J. From birth to death: the complex lives of eukaryotic mRNAs. Science 309, 1514–1518 (2005).
Beltran, M. et al. A natural antisense transcript regulates Zeb2/Sip1 gene expression during Snail1-induced epithelial–mesenchymal transition. Genes Dev. 22, 756–769 (2008). Provides an example of a NAT lncRNA that regulates splicing of the sense encoded mRNA, but with a twist in that the NAT increases protein levels of its target by preventing the splicing of a 5′ UTR IRES-containing intron.
Hastings, M. L., Milcarek, C., Martincic, K., Peterson, M. L. & Munroe, S. H. Expression of the thyroid hormone receptor gene, erbAα, in B lymphocytes: alternative mRNA processing is independent of differentiation but correlates with antisense RNA levels. Nucleic Acids Res. 25, 4296–4300 (1997).
Krystal, G. W., Armstrong, B. C. & Battey, J. F. N-myc mRNA forms an RNA–RNA duplex with endogenous antisense transcripts. Mol. Cell. Biol. 10, 4180–4191 (1990).
Munroe, S. H. & Lazar, M. A. Inhibition of c-erbA mRNA splicing by a naturally occurring antisense RNA. J. Biol. Chem. 266, 22083–22086 (1991).
Faghihi, M. A. & Wahlestedt, C. Regulatory roles of natural antisense transcripts. Nature Rev. Mol. Cell. Biol. 10, 637–643 (2009).
Tripathi, V. et al. The nuclear-retained noncoding RNA MALAT1 regulates alternative splicing by modulating SR splicing factor phosphorylation. Mol. Cell 39, 925–938 (2010).
Hundley, H. A. & Bass, B. L. ADAR editing in double-stranded UTRs and other noncoding RNA sequences. Trends Biochem. Sci. 35, 377–383 (2010).
Peters, N. T., Rohrbach, J. A., Zalewski, B. A., Byrkett, C. M. & Vaughn, J. C. RNA editing and regulation of Drosophila 4f-rnp expression by sas-10 antisense readthrough mRNA transcripts. RNA 9, 698–710 (2003).
Katayama, S. et al. Antisense transcription in the mammalian transcriptome. Science 309, 1564–1566 (2005).
Carrieri, C. et al. Long non-coding antisense RNA controls Uchl1 translation through an embedded SINEB2 repeat. Nature 491, 454–457 (2012). Discovers and characterizes the first lncRNA of a potentially new class of partially antisense SINE2B repeat-containing lncRNAs which upregulates translation of targets.
Nishizawa, M. et al. Nutrient-regulated antisense and intragenic RNAs modulate a signal transduction pathway in yeast. PLoS Biol. 6, 2817–2830 (2008).
Gong, C. & Maquat, L. E. lncRNAs transactivate STAU1-mediated mRNA decay by duplexing with 3′ UTRs via Alu elements. Nature 470, 284–288 (2011). Provides the first evidence that Alu element-containing lncRNAs can transactivate SMD by imperfectly base pairing with 3′ UTR Alu elements in target mRNAs.
Faghihi, M. A. et al. Expression of a noncoding RNA is elevated in Alzheimer's disease and drives rapid feed-forward regulation of β-secretase. Nature Med. 14, 723–730 (2008).
Faghihi, M. A. et al. Evidence for natural antisense transcript-mediated inhibition of microRNA function. Genome Biol. 11, R56 (2010).
Salmena, L., Poliseno, L., Tay, Y., Kats, L. & Pandolfi, P. P. A ceRNA hypothesis: the Rosetta Stone of a hidden RNA language? Cell 146, 353–358 (2011).
Cesana, M. et al. A long noncoding RNA controls muscle differentiation by functioning as a competing endogenous RNA. Cell 147, 358–369 (2011).
Wang, J. et al. CREB up-regulates long non-coding RNA, HULC expression through interaction with microRNA-372 in liver cancer. Nucleic Acids Res. 38, 5366–5383 (2010).
Franco-Zorrilla, J. M. et al. Target mimicry provides a new mechanism for regulation of microRNA activity. Nature Genet. 39, 1033–1037 (2007).
Hansen, T. B. et al. Natural RNA circles function as efficient microRNA sponges. Nature http://dx.doi.org/10.1038/nature11993 (2013). References 77 and 78 provide powerful evidence that circRNAs, covalently linked by the head-to-tail splicing of exons, can function as miRNA sponges to suppress miRNA activity.
Memczak, S. et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature http://dx.doi.org/10.1038/nature11928 (2013).
Poliseno, L. et al. A coding-independent function of gene and pseudogene mRNAs regulates tumour biology. Nature 465, 1033–1038 (2010).
Yin, Q.-F. et al. Long noncoding RNAs with snoRNA ends. Mol. Cell 48, 219–230 (2012). Describes the discovery of a new class of intron-derived lncRNAs flanked by snoRNAs and shows that one in particular associates with splicing regulators to alter splicing patterns.
Hellwig, S. & Bass, B. L. A starvation-induced noncoding RNA modulates expression of Dicer-regulated genes. Proc. Natl Acad. Sci. USA 105, 12897–12902 (2008).
Pijlman, G. P. et al. A highly structured, nuclease-resistant, noncoding RNA produced by flaviviruses is required for pathogenicity. Cell Host Microbe 4, 579–591 (2008).
Moon, S. L. et al. A noncoding RNA produced by arthropod-borne flaviviruses inhibits the cellular exoribonuclease XRN1 and alters host mRNA stability. RNA 18, 2029–2040 (2012).
Liu, X., Li, D., Zhang, W., Guo, M. & Zhan, Q. Long non-coding RNA gadd7 interacts with TDP-43 and regulates Cdk6 mRNA decay. EMBO J. 31, 4415–4427 (2012).
Buratti, E. & Baralle, F. E. The multiple roles of TDP-43 in pre-mRNA processing and gene expression regulation. RNA Biol. 7, 420–429 (2010).
Colombrita, C. et al. TDP-43 and FUS RNA-binding proteins bind distinct sets of cytoplasmic messenger RNAs and differently regulate their post-transcriptional fate in motoneuron-like cells. J. Biol. Chem. 287, 15635–15647 (2012).
Strong, M. J. et al. TDP43 is a human low molecular weight neurofilament (hNFL) mRNA-binding protein. Mol. Cell. Neurosci. 35, 320–327 (2007).
Volkening, K., Leystra-Lantz, C., Yang, W., Jaffee, H. & Strong, M. J. Tar DNA binding protein of 43 kDa (TDP-43), 14-3-3 proteins and copper/zinc superoxide dismutase (SOD1) interact to modulate NFL mRNA stability. Implications for altered RNA processing in amyotrophic lateral sclerosis (ALS). Brain Res. 1305, 168–182 (2009).
Tollervey, J. R. et al. Characterizing the RNA targets and position-dependent splicing regulation by TDP-43. Nature Neurosci. 14, 452–458 (2011).
Zappulla, D. C. & Cech, T. R. Yeast telomerase RNA: a flexible scaffold for protein subunits. Proc. Natl Acad. Sci. USA 101, 10024–10029 (2004).
Halic, M. et al. Structure of the signal recognition particle interacting with the elongation-arrested ribosome. Nature 427, 808–814 (2004).
Pfeiffer, V. & Lingner, J. TERRA promotes telomere shortening through exonuclease 1-mediated resection of chromosome ends. PLoS Genet. 8, e1002747 (2012).
Carmo-Fonseca, M. & Rino, J. RNA seeds nuclear bodies. Nature Cell Biol. 13, 110–112 (2011).
Shevtsov, S. P. & Dundr, M. Nucleation of nuclear bodies by RNA. Nature Cell Biol. 13, 167–173 (2011).
Sunwoo, H. et al. MEN ε/β nuclear-retained non-coding RNAs are up-regulated upon muscle differentiation and are essential components of paraspeckles. Genome Res. 19, 347–359 (2009).
Sasaki, Y. T. F., Ideue, T., Sano, M., Mituyama, T. & Hirose, T. MENε/β noncoding RNAs are essential for structural integrity of nuclear paraspeckles. Proc. Natl Acad. Sci. USA 106, 2525–2530 (2009).
Mao, Y. S., Sunwoo, H., Zhang, B. & Spector, D. L. Direct visualization of the co-transcriptional assembly of a nuclear body by noncoding RNAs. Nature Cell Biol. 13, 95–101 (2011). Uses a live-cell imaging system to directly visualize paraspeckle protein recruitment and shows that NEAT1 lncRNA transcription regulates paraspeckle maintenance with the lncRNA potentially acting as an assembly platform.
Cieśla, J. Metabolic enzymes that bind RNA: yet another level of cellular regulatory network? Acta Biochim. Pol. 53, 11–32 (2006).
Hentze, M. W. & Argos, P. Homology between IRE-BP, a regulatory RNA-binding protein, aconitase, and isopropylmalate isomerase. Nucleic Acids Res. 19, 1739–1740 (1991).
Mukhopadhyay, R., Jia, J., Arif, A., Ray, P. S. & Fox, P. L. The GAIT system: a gatekeeper of inflammatory gene expression. Trends Biochem. Sci. 34, 324–331 (2009).
Lotvall, J. & Valadi, H. Cell to cell signalling via exosomes through esRNA. Cell Adh. Migr. 1, 156–158 (2007).
Ramachandran, S. & Palanisamy, V. Horizontal transfer of RNAs: exosomes as mediators of intercellular communication. Wiley Interdiscip. Rev. RNA 3, 286–293 (2012).
Valadi, H. et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nature Cell Biol. 9, 654–659 (2007).
Skog, J. et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nature Cell Biol. 10, 1470–1476 (2008).
Mathivanan, S., Fahner, C. J., Reid, G. E. & Simpson, R. J. ExoCarta 2012: database of exosomal proteins, RNA and lipids. Nucleic Acids Res. 40, D1241–D1244 (2012).
Huang, X. et al. Characterization of human plasma-derived exosomal RNAs by deep sequencing. BMC Genomics 14, 319 (2013). Characterizes, for the first time, the RNA content of exosomes by RNA sequencing and reveals that lncRNAs are indeed present in these membrane-bound vesicles.
Yu, K., Chedin, F., Hsieh, C.-L., Wilson, T. E. & Lieber, M. R. R-loops at immunoglobulin class switch regions in the chromosomes of stimulated B cells. Nature Immunol. 4, 442–451 (2003).
Abarrategui, I. & Krangel, M. S. Noncoding transcription controls downstream promoters to regulate T-cell receptor-α recombination. EMBO J. 26, 4380–4390 (2007).
Pone, E. J., Xu, Z., White, C. A., Zan, H. & Casali, P. B cell TLRs and induction of immunoglobulin class-switch DNA recombination. Front. Biosci. 17, 2594–2615 (2012).
Stavnezer, J. & Amemiya, C. T. Evolution of isotype switching. Semin. Immunol. 16, 257–275 (2004).
Hackney, J. A. et al. DNA targets of AID evolutionary link between antibody somatic hypermutation and class switch recombination. Adv. Immunol. 101, 163–189 (2009).
Selsing, E. Ig class switching: targeting the recombinational mechanism. Curr. Opin. Immunol. 18, 249–254 (2006).
Abarrategui, I. & Krangel, M. S. Regulation of T cell receptor-α gene recombination by transcription. Nature Immunol. 7, 1109–1115 (2006).
Cobb, R. M., Oestreich, K. J., Osipovich, O. A. & Oltz, E. M. Accessibility control of V(D)J recombination. Adv. Immunol. 91, 45–109 (2006).
Petes, T. D. Meiotic recombination hot spots and cold spots. Nature Rev. Genet. 2, 360–369 (2001).
Wahls, W. P., Siegel, E. R. & Davidson, M. K. Meiotic recombination hotspots of fission yeast are directed to loci that express non-coding RNA. PLoS ONE 3, e2887 (2008).
Spitale, R. C. et al. RNA SHAPE analysis in living cells. Nature Chem. Biol. 9, 18–20 (2013).
Martin, L. et al. Systematic reconstruction of RNA functional motifs with high-throughput microfluidics. Nature Methods 9, 1192–1194 (2012).
Paige, J. S., Wu, K. Y. & Jaffrey, S. R. RNA mimics of green fluorescent protein. Science 333, 642–646 (2011).
Tsai, M.-C., Spitale, R. C. & Chang, H. Y. Long intergenic noncoding RNAs: new links in cancer progression. Cancer Res. 71, 3–7 (2011).
Zhao, J., Sun, B. K., Erwin, J. A., Song, J. J. & Lee, J. T. Polycomb proteins targeted by a short repeat RNA to the mouse X chromosome. Science 322, 750–756 (2008).
Rinn, J. L. et al. Functional demarcation of active and silent chromatin domains in human HOX loci by noncoding RNAs. Cell 129, 1311–1323 (2007).
Wang, K. C. et al. A long noncoding RNA maintains active chromatin to coordinate homeotic gene expression. Nature 472, 120–124 (2011).
Acknowledgements
The authors are most grateful to T. Nilsen and K. Baker for insights and suggestions. The authors regret that not all contributions of their colleagues could be discussed due to space constraints. Work in the authors' laboratory is funded by the National Institute of General Medical Sciences (NIGMS) (GM080465).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Chromatin
-
Condensed DNA structure that is associated with histone proteins and other DNA-binding proteins.
- Transcription activator-like effectors
-
(TALEs). Naturally found in some bacteria, TALEs are proteins that bind DNA through repeat domains, and their code for sequence specificity has been elucidated allowing sequence specific TALEs to be engineered.
- PUF proteins
-
A family of sequence-specific RNA-binding proteins, which bind 3′ untranslated regions within mRNAs to repress target mRNA translation.
- Pseudogenes
-
Dysfunctional relatives of normal genes thought to arise from duplication or retrotransposition.
- Chromatin-modifying complexes
-
Protein complexes that catalyse the covalent chemical modification of chromatin.
- Enhancers
-
Short regions of DNA that enhance the expression of genes at varying distances. Effects can be mediated by transcription factor binding to these sites.
- Alu SINE elements
-
Highly abundant retrotransposons of the short interspersed nuclear elements (SINE) family.
- Nuclear subdomains
-
Non-membrane bound subcompartments of eukaryotic nuclei where factors with similar functions colocalize.
- GAL locus
-
An inducible locus in yeast comprising the GAL1 and GAL10 genes, which are required for galactose metabolism.
- Alternative splicing
-
An mRNA processing step whereby exons can be alternatively used to generate different isoforms of the same gene.
- Internal ribosome entry sites
-
(IRESs). Nucleotide sequence that allows cap-independent translation initiation within the middle of an mRNA transcript.
- Spliceosome
-
The macromolecular machinery (composed of both RNA and protein) responsible for pre-mRNA splicing.
- miRNAs
-
(miRNAs). A class of short (∼23 nucleotides) endogenous non-coding RNAs that control gene expression post-transcriptionally through either translational repression or mRNA degradation.
- Competing endogeneous RNA
-
(ceRNA). RNA transcripts (both coding and non-coding), which share microRNA-targeting sites and thus regulate each other via direct competition for microRNA binding.
- Circular RNA
-
(circRNA). As opposed to conventional linear RNA transcripts, the 5′ and 3′ ends of circular RNAs are covalently linked together.
- Small nucleolar RNA
-
(snoRNA). A class of small RNA molecules that guide the chemical modification of other RNA transcripts.
- sno-lncRNAs
-
(small nucleolar long non-coding RNAs). Class of intron-derived long non-coding RNA flanked by snoRNA ends.
- Dicer
-
An RNase III family endoribonuclease responsible for the processing of pre-miRNAs into short double-stranded RNAs to be loaded into the RNA-induced silencing (RISC) complex.
- Adaptive immune system
-
A system of specialized cells that create immunological memory via specific antibodies after an initial response to a pathogen.
Rights and permissions
About this article
Cite this article
Geisler, S., Coller, J. RNA in unexpected places: long non-coding RNA functions in diverse cellular contexts. Nat Rev Mol Cell Biol 14, 699–712 (2013). https://doi.org/10.1038/nrm3679
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3679
This article is cited by
-
Expression level and clinical significance of NBAT-1 in human cancers: a systematic review and meta-analysis
BMC Cancer (2024)
-
LINC00571 drives tricarboxylic acid cycle metabolism in triple-negative breast cancer through HNRNPK/ILF2/IDH2 axis
Journal of Experimental & Clinical Cancer Research (2024)
-
Unveiling dysregulated lncRNAs and networks in non-syndromic cleft lip with or without cleft palate pathogenesis
Scientific Reports (2024)
-
Construction of a New Immune-Related Competing Endogenous RNA Network with Prognostic Value in Lung Adenocarcinoma
Molecular Biotechnology (2024)
-
SNHG14 Elevates NFAT5 Expression Through Sequestering miR-375-3p to Promote MPP + -Induced Neuronal Apoptosis, Inflammation, and Oxidative Stress in Parkinson’s Disease
Neurochemical Research (2024)