Abstract
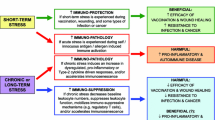
The adaptive responses of the body to challenges, often known as “stressors”, consists of active responses that maintain homeostasis. This process of adaptation is known as “allostasis”, meaning “achieving stability through change”. Many systems of the body show allostasis, including the autonomic nervous system and hypothalamo-pituitary-adrenal (HPA) axis and they help to re-establish or maintain homeostasis through adaptation. The brain also shows allostasis, involving the activation of nerve cell activity and the release of neurotransmitters. When the individual is challenged repeatedly or when the allostatic systems remain turned on when no longer needed, the mediators of allostasis can produce a wear and tear on the body that has been termed “allostatic load”. Examples of allostatic load include the accumulation of abdominal fat, the loss of bone minerals and the atrophy of nerve cells in the hippocampus. Circulating stress hormones play a key role, and, in the hippocampus, excitatory amino acids and NMDA receptors are important mediators of neuronal atrophy. The aging brain seems to be more vulnerable to such effects, although there are considerable individual differences in vulnerability that can be developmentally determined. Yet, at the same time, excitatory amino acids and NMDA receptors mediate important types of plasticity in the hippocampus. Moreover, the brain retains considerable resilience in the face of stress, and estrogens appear to play a role in this resilience. This review discusses the current status of work on underlying mechanisms for these effects.
Similar content being viewed by others
REFERENCES
Sterling, P. and Eyer, J. 1988. Allostasis: A New Paradigm to Explain Arousal Pathology. In Fisher, S., and J. Reason, eds. Handbook of Life Stress, Cognition and Health. New York, John Wiley & Sons., 629-649.
McEwen, B. S. and Stellar, E. 1993. Stress and the Individual: Mechanisms leading to disease. Archives of Internal Medicine 153:2093-2101.
McEwen, B. S. 1998. Protective and Damaging Effects of Stress Mediators. New England J. Med. 338:171-179.
Seeman, T. E., Singer, B. H., Rowe, J. W., Horwitz, R. I., and McEwen, B. S. 1997. Price of adaptation-allostatic load and its health consequences: MacArthur studies of successful aging. Arch. Intern. Med. 157:2259-2268.
Landfield, P., Waymire, J., and Lynch, G. 1978. Hippocampal aging and adrenocorticoids: quantitative correlation. Science 202:1098-1101.
Landfield, P. 1987. Modulation of brain aging correlates by long-term alterations of adrenal steroids and neurally-active peptides. Prob. Brain Res. 72:279-300.
Sapolsky, R. 1992. Stress, the Aging Brain and the Mechanisms of Neuron Death. Cambridge MIT Press 1:423.
Lupien, S., Lecours, A. R., Lussier, I., Schwartz, G., Nair, N. P. V., and Meaney, M. J. 1994. Basal cortisol levels and cognitive deficits in human aging. J. Neurosci. 14:2893-2903.
Seeman, T. E., McEwen, B. S., Singer, B. H., Albert, M. S., and Rowe, J. W. 1997. Increase in Urinary Cortisol Excretion and Memory Declines: MacArthur Studies of Successful Aging. J. Clin. Endocr. Metab. 82:2458-2465.
Lupien, S. J., DeLeon, M. J., De Santi, S., Convit, A., Tarshish, C., Nair, N. P. V., Thakur, M., McEwen, B. S., Hauger, R. L., and Meaney, M. J. 1998. Cortisol levels during human aging predict hippocampal atrophy and memory deficits. Nature Neuroscience 1:69-73.
Eichenbaum, H. 1997. How does the brain organize memories? Science 277:330-332.
DeKloet, E. R., Vreugdenhil, E., Oitzl, M. S., and Joels, M. 1998. Brain corticosteroid receptor balance in health and disease. Endocr. Rev. 19:269-301.
Sapolsky, R., Krey, L., and McEwen, B. S. 1986. The neuroendocrinology of stress and aging: the glucocorticoid cascade hypothesis. Endocr. Rev. 7:284-301.
McEwen, B. S., Albeck, D., Cameron, H., Chao, H. M., Gould, E., Hastings, N., Kuroda, Y., Luine, V., Magarinos, A. M., McKittrick, C. R., Orchinik, M., Pavlides, C., Vaher, P., Watanabe, Y., and Weiland, N. 1995. Stress and the Brain: A Paradoxical Role for Adrenal Steroids. In Litwack, G. D., ed. Vitamins And Hormones. Academic Press, Inc., 371-402.
Cameron, H. A. and Gould, E. 1996. The Control of Neuronal Birth and Survival. In Shaw, C. A., ed. Receptor Dynamics in Neural Development. New York, CRC Press, 141-157.
McEwen, B. S. 1999. Stress and hippocampal plasticity. Annu. Rev. Neurosci. 22:105-122.
McEwen, B. S. and Alves, S. H. 1999. Estrogen Actions in the Central Nervous System. Endocr. Rev. 20:279-307.
Landfield, P. W. and Eldridge, J. C. 1994. Evolving aspects of the glucocorticoid hypothesis of brain aging: Hormonal modulation of neuronal calcium homeostasis. Neurobiol. Aging 15:579-588.
Porter, N. M., Thibault, O., Thibault, V., Chen, K.-C., and Land-field, P. W. 1997. Calcium channel density and hippocampal cell death with age in long-term culture. J. Neurosci. 17:5629-5639.
Norris, C. M., Halpain, S., and Foster, T. C. 1998. Reversal of age-related alterations in synaptic plasticity by blockade of Ltype Ca2+channels. J. Neurosci 18:3171-3179.
Maren, S. 1995. Properties and mechanisms of long-term synaptic plasticity in the mammalian brain: relationships to learning and memory. Neurobiol. Learning & Mem. 63:1-18.
Joels, M. 1997. Steroid hormones and excitability in the mammalian brain. Frontiers in Neuroendocrinol. 18:2-48.
Kerr, S., Campbell, L., Applegate, M., Brodish, A., and Land-field, P. 1991. Chronic stress-induced acceleration of electrophysiologic and morphometric biomarkers of hippocampal aging. J. Neurosci. 11:1316-1324.
McCord, J. 1985. Oxygen-derived free radicals in postischemic tissue injury. New Engl. J. Med. 312:159-163.
Liu, J., Fischer, A., Amiri, N., Timiras, P. S., and Ames, B. N. 1998. Stress-induced oxidative damage in the brain: stress hormones enhance, but an antistress hormone (DHEA), inhibits, oxidative damage and beta-amyloid production in cell culture. Oxygen Society Meeting Poster.
Rasmussen, T., Schliemann, T., Sorensen, J. C., Zimmer, J., and West, M. J. 1996. Memory Impaired Aged Rats: No loss of Principal Hippocampal and Subicular Neurons. Neurobiol. Aging 14:143-147.
Rapp, P. R. and Gallagher, M. 1996. Preserved neuron number in the hippocampus of aged rats with spatial learning deficits. Proc. Natl. Acad. Sci. USA 93:9926-9930.
McEwen, B. S. 1999. Stress and the Aging Hippocampus. Frontiers in Neuroendocrinol. 20:49-70.
Sugaya, K., Chouinard, M., Greene, R., Robbins, M., Personett, D., Kent, C., Gallagher, M., and McKinney, M. 1996. Molecular Indices of Neuronal and Glial Plasticity in the Hippocampal Formation in a Rodent Model of Age-Induced Spatial Learning Impairment. J. Neurosci. 16:3427-3443.
Chan, P. H. 1996. Role of oxidants in ischemic brain damage. Stroke 27:1124-1129.
Lowy, M. T., Gault, L., and Yamamoto, B. K. 1993. Adrenalectomy Attenuates Stress-Induced Elevations in Extracellular Glutamate Concentrations in the Hippocampus. J. Neurochem. 61:1957-1960.
Moghaddam, B., Boliano, M. L., Stein-Behrens, B., and Sapolsky, R. 1994. Glucocorticoids mediate the stress-induced extracellular accumulation of glutamate. Brain Res. 655:251-254.
Schaasfoort, E., DeBrin, L., and Korf, J. 1988. Mild stress stimulates rat hippocampal glucose utilization transiently via NMDA receptors as assessed by lactography. Brain Res. 575:58-63.
De Bruin, L. A., Schasfoort, M. C., Stefens, A. B., and Korf, J. 1994. Effects of stress and exercise on rat hippocampus and striatum extracellular lactate. Am. J. Physiol. 259:R773-R779.
Lowy, M. T., Wittenberg, L., and Yamamoto, B. K. 1995. Effect of Acute Stress on Hippocampal Glutamate Levels and Spectrin Proteolysis in Young and Aged Rats. J. Neurochem. 65:268-274.
Reagan, L. P., and McEwen, B. S. 1997. Controversies surrounding glucocorticoid-mediated cell death in the hippocampus. J. Chem. Neuroanat. 13:149-167.
Keller, J. N. and Mattson, M. P. 1998. Roles of lipid peroxidation in modulation of cellular signaling pathways, cell dysfunction, and death in the nervous system. Reviews in Neurosci. 9:105-116.
Maren, S. 1999. Long-term potentiation in the amygdala: a mechanism for emotional learning and memory. TINS 22:561-567.
Bliss, T. V. P. and Collingridge, G. L. 1993. A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361:31-39.
Choi, D. 1988. Calcium-mediated neurotoxicity: relationship to specific channel types and role in ischemic damage. TINS 11: 465-469.
Weiland, N. G., Orchinik, M., and Tanapat, P. 1997. Chronic corticosterone treatment induces parallel changes in N-methyl-Daspartate receptor subunit messenger RNA levels and antagonist binding sites in the hippocampus. Neuroscience 78:653-662.
Bartanusz, V., Aubry, J. M., Pagliusi, S., Jezova, D., Baffi, J., and Kiss, J. Z. 1995. Stress-Induced Changes in Messenger RNA Levels of N-Methyl-D-Aspartate and Ampa Receptor Subunits in Selected Regions of the Rat Hippocampus and Hypothalamus. Neuroscience 66:247-252.
Weinstock, M., Poltyrev, T., Schorer-Apelbaum, D., Men, D., and McCarty, R. 1998. Effect of prenatal stress on plasma corticosterone and catecholamines in response to footshock in rats. Physiol. & Behav. 64:439-444.
Ader, R. 1968. Effects of early experiences on emotional and physiological reactivity in the rat. J. Comp. Physiol. Psych. 66: 264-268.
Hess, J. L., Denenberg, V. H., Zarrow, M. X., and Pfeifer, W. D. 1968. Modification of the corticosterone response curve as a function of handling in infancy. Physiol. & Behav. 4: 109-111.
Levine, S., Haltmeyer, G., Kara, G., and Denenberg, V. 1967. Physiological and behavioral effects of infantile stimulation. Physiol. Behav. 2:55-59.
Dellu, F., Mayo, W., Vallee, M., LeMoal, M., and Simon, H. 1994. Reactivity to novelty during youth as a predictive factor of cognitive impairment in the elderly: a longitudinal study in rats. Brain Res. 653:51-56.
Piazza, P. V., Marinelli, M., Jodogne, C., Deroche, V., Rouge-Pont, F., Maccari, S., Le Moal, M., and Simon, H. 1994. Inhibition of corticosterone synthesis by Metrapone decreases cocaine-induced locomotion and relapse of cocaine self-administration. Brain Res. 658:259-264.
Deroche, V., Piazza, P. V., LeMoal, M., and Simon, H. 1993. Individual differences in the psychomotor effects of morphine are predicted by reactivity to novelty and influenced by corticosterone secretion. Brain Res. 623:341-344.
Meaney, M., Aitken, D., Berkel, H., Bhatnager, S., and Sapolsky, R. 1988. Effect of neonatal handling of age-related impairments associated with the hippocampus. Science 239:766-768.
Catalani, A., Marinelli, M., Scaccianoce, S., Nicolai, R., Muscolo, L. A. A., Porcu, A., Koranyi, L., Piazza, P. V., and Angelucci, L. 1993. Progeny of mothers drinking corticosterone during lactation has lower stress-induced corticosterone secretion and better cognitive performance. Brain Res. 624:209-215.
Meaney, M. J., Tannenbaum, B., Francis, D., Bhatnagar, S., Shanks, N., Viau, V., O'Donnell, D., and Plotsky, P. M. 1994. Early environmental programming hypothalamic-pituitary-adrenal responses to stress. Seminars in Neurosci. 6:247-259.
Pavlides, C., Kimura, A., Magarinos, A. M., and McEwen, B. S. 1995. Hippocampal homosynaptic long-term depression/ depotentiation induced by adrenal steroids. Neuroscience 68:379-385.
Pavlides, C., Watanabe, Y., Magarinos, A. M., and McEwen, B. S. 1995. Opposing role of adrenal steroid Type I and Type II receptors in hippocampal long-term potentiation. Neuroscience 68:387-394.
Pavlides, C., Kimura, A., Magarinos, A. M., and McEwen, B. S. 1994. Type I adrenal steroid receptors prolong hippocampal long-term potentiation. NeuroReport 5:2673-2677.
Kerr, D. S., Huggett, A. M., and Abraham, W. C. 1994. Modulation of hippocampal long-term potentiation and long-term depression by corticosteroid receptor activation. Psychobiology 22:123-133.
Barnes, C., McNaughton, B., Goddard, G., Douglas, R., and Adamec, R. 1977. Circadian rhythm of synaptic excitability in rat and monkey central nervous system. Science 197:91-92.
Dana, R. C. and Martinez, J. L. 1984. Effect of adrenalectomy on the circadian rhythm of LTP. Brain Res. 308:392-395.
Diamond, D. M., Bennett, M. C., Fleshner, M., and Rose, G. M. 1992. Inverted-U relationship between the level of peripheral corticosterone and the magnitude of hippocampal primed burst potentiation. Hippocampus 2:421-430.
Diamond, D. M., Fleshner, M., Ingersoll, N., and Rose, G. M. 1996. Psychological stress impairs spatial working memory: relevance to electrophysiological studies of hippocampal function. Behav. Neurosci. 110:661-672.
Roozendaal, B. and McGaugh, J. L. 1997. Glucocorticoid receptor agonist and antagonist administration into the basolateral but not central amygdala modulates memory storage. Neurobiol. Learning & Mem. 67:176-179.
Corodimas, K. P., LeDoux, J. E., Gold, P. W., and Schulkin, J. 1994. Corticosterone potentiation of learned fear. Annals of the New York Acad. Sci. 746:392.
Diamond, D. M., Fleshner, M., and Rose, G. M. 1994. Psychological stress repeatedly blocks hippocampal primed burst potentiation in behaving rats. Behav. Brain Research 62:1-9.
deQuervain, D. J. F., Roozendaal, B., and McGaugh, J. L. 1998. Stress and glucocorticoids impair retrieval of long-term spatial memory. Nature 394:787-790.
Galea, L. A. M., Tanapat, P., and Gould, E. 1996. Exposure to predator odor suppresses cell proliferation in the dentate gyrus of adult rats via a cholinergic mechanism. Abstract, Soc. Neurosci. 22:#474.8, p.1196.
Ikegaya, Y., Saito, H., and Abe, K. 1997. The basomedial and basolateral amygdaloid nuclei contribute to the induction of long-term potentiation in the dentate gyrus in vivo. Eur. J. Neurosci. 8:1833-1839.
LeDoux, J. E. 1995. In search of an emotional system in the brain: leaping from fear to emotion and consciousness. In Gazzaniga, M., ed. The Cognitive Neurosciences. Cambridge, MIT Press, 1049-1061.
Woolley, C., Gould, E., Frankfurt, M., and McEwen, B. S. 1990. Naturally occurring fluctuation in dendritic spine density on adult hippocampal pyramidal neurons. J. Neurosci. 10:4035-4039.
Popov, V. I. and Bocharova, L. S. 1992. Hibernation-induced structural changes in synaptic contacts between mossy fibres and hippocampal pyramidal neurons. Neuroscience 48:53-62.
Popov, V. I., Bocharova, L. S., and Bragin, A. G. 1992. Repeated changes of dendritic morphology in the hippocampus of ground squirrels in the course of hibernation. Neuroscience 48:45-51.
McEwen, B. S. and Woolley, C. S. 1994. Estradiol and progesterone regulate neuronal structure and synaptic connectivity in adult as well as developing brain. Exp. Geront. 29:431-436.
McEwen, B. S., Gould, E., Orchinik, M., Weiland, N. G., and Woolley, C. S. 1995. Oestrogens and the structural and functional plasticity of neurons: implications for memory, aging and neurodegenerative processes. In Goode, J., ed. Ciba Foundation Symposium #191 The Non-reproductive Actions of Sex Steroids. London, CIBA Foundation, 52-73.
Cameron, H. A. and Gould, E. 1994. Adult neurogenesis is regulated by adrenal steroids in the dentate gyrus. Neuroscience 61:203-209.
Woolley, C. and McEwen, B. S. 1994. Estradiol regulates hippocampal dendritic spine density via an N-methyl-D-aspartate receptor dependent mechanism. J. Neurosci. 14:7680-7687.
Magarinos, A. M. and McEwen, B. S. 1995. Stress-induced atrophy of apical dendrites of hippocampal CA3c neurons: involvement of glucocorticoid secretion and excitatory amino acid receptors. Neuroscience 69:89-98.
Weiland, N. G. 1992. Estradiol Selectively Regulates Agonist Binding Sites on the N-Methyl-D-Aspartate Receptor Complex in the CA1 Region of the Hippocampus. Endocrinology 131: 662-668.
Gazzaley, A. H., Benson, D. L., Huntley, G. W., and Morrison, J. H. 1997. Differential subcellular regulation of NMDAR1 protein and mRNA in dendrites of dentate gyrus granule cells after perforant path transection. J. Neurosci. 17:2006-2017.
Gould, E. 1999. Serotonin and hippocampal neurogenesis. Neuropsychopharmacology 21:46S-51S.
van Praag, H., Kempermann, G., and Gage, F. H. 1999. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nature Neurosci. 2:266-270
Kempermann, G., Kuhn, H. G., and Gage, F. H. 1997. More hippocampal neurons in adult mice living in an enriched environment. Nature 586:493-495.
Conrad, C. D., Magarinos, A. M., LeDoux, J. E., and McEwen, B. S. 1999. Repeated restraint stress facilitates fear conditioning independently of causing hippocampal CA3 dendritic atrophy. Behav. Neurosci. 113:1-12.
Eldridge, J. C., Brodish, A., Kute, T. E., and Landfield, P. W. 1989. Apparent age-related resistance of type II hippocampal corticosteroid receptors to down-regulation during chronic escape training. J. Neurosci. 9:3237-3242.
Cameron, H. A. and McKay, D. G. 1999. Restoring production of hippocampal neurons in old age. Nature Neurosci. 2:894-858.
Tanapat, P., Hastings, N. B., Reeves, A. J., and Gould, E. 1999. Estrogen stimulates a transient increase in the number of new neurons in the dentate gyrus of the adult female rat. J. Neurosci. 19:5792-5801.
Galea, L. A. M., McEwen, B. S., Tanapat, P., Deak, T., Spencer, R. L., and Dhabhar, F. S. 1997. Sex differences in dendritic atrophy of CA3 pyramidal neurons in response to chronic restraint stress. Neuroscience 81:689-697.
Gould, E., Westlind-Danielsson, A., Frankfurt, M., and McEwen, B. S. 1990. Sex differences and thyroid hormone sensitivity of hippocampal pyramidal neurons. J. Neurosci. 10: 996-1003.
Williams, C. L. and Meck, W. H. 1991. The organizational effects of gonadal steroids on sexually dimorphic spatial ability. Psychoneuroendocrinology 16:155-176.
Juraska, J. M. 1991. Sex differences in "cognitive" regions of the rat brain. Psychoneuroendocrinology 16:105-119.
Roof, R. L. 1993. The dentate gyrus is sexually dimorphic in prepubescent rats: testosterone plays a significant role. Brain Res. 610:148-151.
Van Cauter, E., Leproult, R., and Kupfer, D. J. 1996. Effects of Gender and Age on the Levels and Circadian Rhythmicity of Plasma Cortisol. J. Clin. Endocrinol. & Metabol. 81:2468-2473.
Komesaroff, P. A., Esler, M. D., and Sudhir, K. 1999. Estrogen supplementation attenuates glucocorticoid and catecholamine responses to mental stress in perimenopausal women. J. Clin. Endocrinol. & Metab. 84:606-610
Goodman, Y., Bruce, A. J., Cheng, B., and Mattson, M. P. 1996. Estrogens attenuate and corticosterone exacerbates excitotoxicity, oxidative injury, and amyloid β-peptide toxicity in hippocampal neurons. J. Neurochem. 66:1836-1844.
Hawk, T., Zhang, Y.-Q., Rajakumar, G., Day, A. L., and Simpkins, J. W. 1998. Testosterone increases and estradiol decreases middle cerebral artery occlusion lesion size in male rats. Brain Res. 796:296-298.
Dubal, D. B., Shughrue, P. J., Wilson, M. E., Merchenthaler, I., and Wise, P. M. 1999. Estradiol modulates bcl-2 in cerebral ischemia: A potential role for estrogen receptors. J. Neurosci. 19:6385-6393.
Sapolsky, R. and Pulsinelli, W. 1985. Glucocorticoids potentiate ischemic injury to neurons: therapeutic implications. Science 229:1397-1399.
Uno, H., Ross, T., Else, J., Suleman, M., and Sapolsky, R. 1989. Hippocampal damage associated with prolonged and fatal stress in primates. J. Neurosci 9:1709-1711.
Henderson, V. W. and Paganini-Hill, A. 1994. Oestrogens and Alzheimer's disease. Ann. Prog. Rep. Med. 2:1-21.
Tang, M. X., Jacobs, D., Stern, Y., Marder, K., Schofield, P., Gurland, B., Andrews, H., and Mayeux, R. 1996. Effect of oestrogen during menopause on risk and age at onset of Alzheimer's disease. Lancet 348:429-432.
Kawas, C., Resnick, S., Morrison, A., Brookmeyer, R., Corrada, M., Zonderman, A., Bacal, C., Lingle, D. D., and Metter, E. 1997. A prospective study of estrogen replacement therapy and the risk of developing Alzheimer's disease: the Baltimore Longitudinal Study of Aging. Neurology 48:1517-1521.
Green, P. S., Gridley, K. E., and Simpkins, J. W. 1996. Estradiol protects against β-amyloid (25-35)-induced toxicity in SK-NSH human neuroblastoma cells. Neurosci. Lett. 218:165-168.
Blaylock, R. L. 1999. Neurodegeneration and aging of the central nervous system: Prevention and treatment by phytochemicals and metabolic nutrients. Integrat. Med. 1:117-133
Kuiper, G. G. J. M., Lemmen, J. G., Carlsson, B., Corton, J. C., Safe, S. H., van der Saag, P. T., van der Burg, B., and Gustafsson, J.-A. 1998. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology 139:4252-4263.
Bishop, J. and Simpkins, J. W. 1994. Estradiol treatment increases viability of glioma and neuroblastoma cells in vitro. Mol. & Cell. Neurosci. 5:303-308.
Green, P. S., Bishop, J., and Simpkins, J. W. 1997. 17α-Estradiol exerts neuroprotective effects on SK-N-SH cells. J. Neurosci. 17:511-515.
Mooradian, A. D. 1993. Antioxidant properties of steroids. J. Steroid Biochem. Molec. Biol. 45:509-511.
Behl, C., Skutella, T., Lezoualc'h, F., Post, A., Widmann, M., Newton, C. J., and Holsboer, F. 1997. Neuroprotection against oxidative stress by estrogens: structure-activity relationship. Mole. Pharm. 51:535-541.
Behl, C., Widmann, M., Trapp, T., and Holsboer, F. 1998. 17 beta estradiol protects neurons from oxidative stress-induced cell death in vitro. Biochem. Biophys. Res. Comm. 216:473-482.
Birge, S. J. 1994. The Role of Estrogen Deficiency in the Aging Central Nervous System. In Lobo, R. A., ed. Treatment of the Postmenopausal Woman: Basic and Clinical Aspects. New York, Raven Press, Ltd., 153-157. Notes: Chapter Num: 14.
Henderson, V. W., Paganini-Hill, A., Emanuel, C. K., Dunn, M. E., and Buckwalter, J. G. 1994. Estrogen Replacement Therapy in Older Women: Comparisons between Alzheimer's Disease Cases and Nondemented Control Subjects. Archives of Neurology 51:896-900.
Paganini-Hill, A. and Henderson, V. W. 1994. Estrogen Deficiency and Risk of Alzheimer's Disease in Women. American Journal of Epidemilogy 3:3-16.
Henderson, V. W., Watt, L., and Buckwalter, J. G. 1996. Cognitive Skills Associated With Estrogen Replacement In Women With Alzheimer's Disease. Psychoneuroendocrinology 12:421-430.
Yaffe, K., Sawaya, G., Lieberburg, I., and Grady, D. 1998. Estrogen therapy in postmenopausal women: Effects on cognitive function and dementia. JAMA 279:688-695.
Joseph, J. A., Shukitt-Hale, B., Denisova, N. A., Prior, R. L., Cao, G., Martin, A., Taglialatela, G., and Bickford, P. C. 1998. Long-term dietary strawberry, spinach, or vitamin E supplementation retards the onset of age-related neuronal signal-transduction and cognitive behavioral deficits. J. Neurosci. 18: 8047-8055.
Le Bars, P. L., Katz, M. M., Berman, N., Itil, T. M., Freedman, A. M., and Schatzberg, A. F. 1997. A placebo-controlled, double-blind, randomized trial of an extract of Ginko biloba for dementia. JAMA 278:1327-1332.
Patisaul, H. B., Whitten, P. L., and Young, L. J. 1999. Regulation of estrogen receptor beta mRNA in the brain: opposite effects of 17β-estradiol and the phytoestrogen, coumestrol. Mol. Brain Res. 67:165-171.
Nagata, C., Takatsuka, N., Inaba, S., Kawakami, N., and Shimizu, H. 1998. Effect of soymilk consumption on serum estrogen concentrations in premenopausal Japanese women. J. Natl. Cancer Inst. 90:1830-1835.
Morrison, R. S., Wenzel, H. J., Kinoshita, Y., Robbins, C. A., Donehower, L. A., and Schwartzkroin, P. A. 1996. Loss of the p53 tumor suppressor gene protects neurons from kainate-induced cell death. J. Neurosci. 16:1337-1345.
Xiang, H., Hochman, D. W., Saya, H., Fujiwara, T., Schwartzkroin, P. A., and Morrison, R. S. 1996. Evidence for p53-mediated modulation of neuronal viability. J. Neurosci. 16:6753-6765.
Kondo, T., Reaume, A. G., Huang, T.-T., Carlson, E., Murakami, K., Chen, S. F., Hoffman, E. K., Scott, R. W., Epstein, C. J., and Chan, P. H. 1997. Reduction of CuZn-superoxide dismutase activity exacerbates neuronal cell injury and edema formation after transient focal cerebral ischemia. J. Neurosci. 17:4180-4189.
Reagan, L. P., Magarinos, A. M., and McEwen, B. S. 1998. Molecular changes induced by stress in streptozotocin (STZ) diabetic rats. Society for Neuroscience Abstract.
Pouliot, W. A., Handa, R. J., and Beck, S. G. 1996. Androgen Modulates N-Methyl-D-Aspartate-mediated Depolarization in CA1 Hippocampal Pyramidal Cells. Synapse 23:10-19.
Grindley, K. E., Green, P. S., and Simpkins, J. W. 1997. Low concentrations of estradiol reduce β-amyloid (25-35) induced toxicity, lipid peroxidation and glucose utilization in human SKN-SH neuroblastoma cells. Brain Res. 778:158-165.
Kimonides, V. G., Khatibi, N. H., Sofroniew, M. V., and Herbert, J. 1998. Dehydroepiandrosterone (DHEA) and DHEA-sulfate (DHEAS) protect hippocampal neurons against excitatory amino acid-induced neurotoxicity. Proc. Nat. Acad. Sci. USA 95:1852-1857.
Bastianetto, S., Ramassamy, C., Poirier, J., and Quirion, R. 1999. Dehydroepiandrosterone (DHEA) protects hippocampal cells from oxidative stress-induced damage. Mol. Brain Res. 66:35-41
Ho, D. Y., Saydam, T. C., Fink, S. L., Lawrence, M. S., and Sapolsky, R. M. 1995. Defective herpes simplex virus vectors expressing the rat brain glucose transporter protect cultured neurons from necrotic insults. J. Neurochem. 65:842-850.
McEwen, B. S. 1999. The molecular and neuroanatomical basis for estrogen effects in the central nervous system. J. Clin. Endocrinol. Metab. 84:1790-1797.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McEwen, B.S. Allostasis, Allostatic Load, and the Aging Nervous System: Role of Excitatory Amino Acids and Excitotoxicity. Neurochem Res 25, 1219–1231 (2000). https://doi.org/10.1023/A:1007687911139
Issue Date:
DOI: https://doi.org/10.1023/A:1007687911139