Abstract
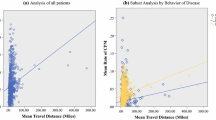
Apart from tumour, treatment and patient characteristics at diagnosis, access to healthcare delivery may as well play a significant role in breast cancer prognosis. This study aimed to assess the additional impact exerted on survival by travel burden—a surrogate indicator of limited access to healthcare- expressed as geographical distance and/or time needed to reach the tertiary healthcare center from the patient’s residence. Between 1997 and 2005, 2,789 women participated in therapeutic clinical trials conducted by the Hellenic Cooperative Oncology Group. The effect of geographical distance and travel time between patient’s residence and treating hospital on survival was estimated using Cox proportional hazards regression adjusting for age, menopausal status, tumour size/grade, positive nodes (number), hormonal receptor status, HER2 overexpression, surgery type/treatment protocol as well as for body mass index >30 kg/m2. More aggressive tumour features, older treatment protocols and modifiable patient characteristics, such as obesity (HR: 1.27) adversely impacted on breast cancer survival. In addition, less studied indicators of access to healthcare, such as geographic distance >350 km and travel time >4 h were independently and significantly associated with worse outcomes (HR = 1.43 and 1.34 respectively). In conclusion, to address inequalities in breast cancer survival, improvements in access to healthcare services related to increased travel burden especially for patients of lower socioeconomic status should be considered, more than ever at times of financial crisis and independently of already known modifiable patient characteristics.

Similar content being viewed by others
References
Soerjomataram I, Louwman MWJ, Ribot JG, Roukema JA, Coebergh JWW. An overview of prognostic factors for long-term survivors of breast cancer. Breast Cancer Res Treat. 2008;107(3):309–30.
Clarke M, Collins R, Darby S, Davies C, Elphinstone P, Evans E, et al. Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;366(9503):2087–106. doi:10.1016/S0140-6736(05)67887-7.
Weigel MT, Dowsett M. Current and emerging biomarkers in breast cancer: prognosis and prediction. Endocr Relat Cancer. 2010;17(4):R245–62. doi:10.1677/erc-10-0136.
Malamou-Mitsi V, Gogas H, Dafni U, Bourli A, Fillipidis T, Sotiropoulou M, et al. Evaluation of the prognostic and predictive value of p53 and Bcl-2 in breast cancer patients participating in a randomized study with dose-dense sequential adjuvant chemotherapy. Ann Oncol. 2006;17(10):1504–11. doi:10.1093/annonc/mdl147.
Dal Maso L, Zucchetto A, Talamini R, Serraino D, Stocco CF, Vercelli M, et al. Effect of obesity and other lifestyle factors on mortality in women with breast cancer. Int J Cancer. 2008;123(9):2188–94.
Hellmann SS, Thygesen LC, Tolstrup JS, Gronbaek M. Modifiable risk factors and survival in women diagnosed with primary breast cancer: results from a prospective cohort study. Eur J Cancer Prevent. 2010;19(5):366–73. doi:10.1097/CEJ.0b013e32833b4828.
Pierce JP, Stefanick ML, Flatt SW, Natarajan L, Sternfeld B, Madlensky L, et al. Greater Survival after breast cancer in physically active women with high vegetable-fruit intake regardless of obesity. J Clin Oncol. 2007;25(17):2345–51. doi:10.1200/jco.2006.08.6819.
Parkin DM, Boyd L. Cancers attributable to overweight and obesity in the UK in 2010. Br J Cancer. 2011;105(S2):S34–7.
Protani M, Coory M, Martin J. Effect of obesity on survival of women with breast cancer: systematic review and meta-analysis. Breast Cancer Res Treat. 2010;123(3):627–35.
Berclaz G, Li S, Price KN, Coates AS, Castiglione-Gertsch M, Rudenstam CM, et al. Body mass index as a prognostic feature in operable breast cancer: the International Breast Cancer Study Group experience. Ann Oncol. 2004;15(6):875–84. doi:10.1093/annonc/mdh222.
Barnett GC, Shah M, Redman K, Easton DF, Ponder BAJ, Pharoah PDP. Risk factors for the incidence of breast cancer: do they affect survival from the disease? J Clin Oncol. 2008;26(20):3310–6. doi:10.1200/jco.2006.10.3168.
Ewertz M, Jensen M-B, Gunnarsdóttir KÁ, Højris I, Jakobsen EH, Nielsen D, et al. Effect of obesity on prognosis after early-stage breast cancer. J Clin Oncol. 2010;29(1):25–31. doi:10.1200/jco.2010.29.7614.
Abrahamson PE, Gammon MD, Lund MJ, Flagg EW, Porter PL, Stevens J, et al. General and abdominal obesity and survival among young women with breast cancer. Cancer Epidemiol Biomarkers Prev. 2006;15(10):1871–7. doi:10.1158/1055-9965.epi-06-0356.
Dignam JJ, Mamounas EP. Obesity and breast cancer prognosis: an expanding body of evidence. Ann Oncol. 2004;15(6):850–1. doi:10.1093/annonc/mdh241.
Bouchardy C, Verkooijen HM, Fioretta G. Social class is an important and independent prognostic factor of breast cancer mortality. Int J Cancer. 2006;119(5):1145–51.
Dalton SO, Ross L, Düring M, Carlsen K, Mortensen PB, Lynch J, et al. Influence of socioeconomic factors on survival after breast cancer—a nationwide cohort study of women diagnosed with breast cancer in Denmark 1983–1999. Int J Cancer. 2007;121(11):2524–31.
Bradley CJ, Given CW, Roberts C. Race, socioeconomic status, and breast cancer treatment and survival. J Natl Cancer Inst. 2002;94(7):490–6. doi:10.1093/jnci/94.7.490.
Yu X. Socioeconomic disparities in breast cancer survival: relation to stage at diagnosis, treatment and race. BMC Cancer. 2009;9(1):364.
McGinnis JM, Williams-Russo P, Knickman JR. The case for more active policy attention to health promotion. Health Aff. 2002;21(2):78–93. doi:10.1377/hlthaff.21.2.78.
Probst J, Laditka S, Wang J-Y, Johnson A. Effects of residence and race on burden of travel for care: cross sectional analysis of the 2001 US National Household Travel Survey. BMC Health Services Res. 2007;7(1):40.
Zgibor JC, Gieraltowski LB, Talbott EO, Fabio A, Sharma RK, Karimi H. The association between driving distance and glycemic control in rural areas. J Diabetes Sci Technol. 2011;5(3):494–500.
Scoggins JF, Fedorenko CR, Donahue SMA, Buchwald D, Blough DK, Ramsey SD. Is distance to provider a barrier to care for medicaid patients with breast, colorectal, or lung cancer? J Rural Health. 2011; Article first published online: 31 Mar 2011. doi:10.1111/j.1748-0361.2011.00371.x.
Dejardin O, Remontet L, Bouvier AM, Danzon A, Tretarre B, Delafosse P, et al. Socioeconomic and geographic determinants of survival of patients with digestive cancer in France. Br J Cancer. 2006;95(7):944–9.
Crawford SM, Sauerzapf V, Haynes R, Zhao H, Forman D, Jones AP. Social and geographical factors affecting access to treatment of lung cancer. Br J Cancer. 2009;101(6):897–901.
Athas WF, Adams-Cameron M, Hunt WC, Amir-Fazli A, Key CR. Travel distance to radiation therapy and receipt of radiotherapy following breast-conserving surgery. J Natl Cancer Inst. 2000;92(3):269–71. doi:10.1093/jnci/92.3.269.
Jones AP, Haynes R, Sauerzapf V, Crawford SM, Zhao H, Forman D. Travel time to hospital and treatment for breast, colon, rectum, lung, ovary and prostate cancer. Eur J Cancer. 2008;44(7):992–9.
Baird G, Flynn R, Baxter G, Donnelly M, Lawrence J. Travel time and cancer care: an example of the inverse care law? Rural Remote Health. 2008;8(4):1003 (Online). Epub Nov 13, 2008.
Fountzilas G, Skarlos D, Dafni U, Gogas H, Briasoulis E, Pectasides D, et al. Postoperative dose-dense sequential chemotherapy with epirubicin, followed by CMF with or without paclitaxel, in patients with high-risk operable breast cancer: a randomized phase III study conducted by the Hellenic Cooperative Oncology Group. Ann Oncol. 2005;16(11):1762–71. doi:10.1093/annonc/mdi366.
Fountzilas G, Dafni U, Gogas H, Linardou H, Kalofonos HP, Briasoulis E, et al. Postoperative dose-dense sequential chemotherapy with epirubicin, paclitaxel and CMF in patients with high-risk breast cancer: safety analysis of the Hellenic Cooperative Oncology Group randomized phase III trial HE 10/00. Ann Oncol. 2008;19(5):853–60. doi:10.1093/annonc/mdm539.
Gogas H, Dafni U, Karina M, Papadimitriou C, Batistatou A, Bobos M, et al. Postoperative dose-dense sequential versus concomitant administration of epirubicin and paclitaxel in patients with node-positive breast cancer: 5-year results of the Hellenic Cooperative Oncology Group HE 10/00 phase III Trial. Breast Cancer Res Treat. 2011;2011 Dec 21. [Epub ahead of print] doi:10.1007/s10549-011-1913-4.
Papadimitriou CA, Papakostas P, Timotheadou E, Aravantinos G, Bamias A, Fountzilas G. Adjuvant dose-dense sequential chemotherapy with epirubicin, CMF and Weekly paclitaxel in patients with resected high-risk breast cancer: a Hellenic Cooperative Oncology Group (HeCOG) Study. Cancer Invest. 2008;26(5):491–8. doi:10.1080/07357900701829785.
Fountzilas G, Pectasides D, Christodoulou C, Timotheadou E, Economopoulos T, Papakostas P, et al. Adjuvant dose-dense sequential chemotherapy with epirubicin, CMF, and weekly docetaxel is feasible and safe in patients with operable breast cancer. Med Oncol. 2006;23(4):479–88.
Acock AC. Working with missing values. J Marriage Fam. 2005;67:1012–28.
Little RJA, Rubin DB. Statistical analysis with missing data. New York: Wiley; 1987.
Meden T, St. John-Larkin C, Hermes D, Sommerschield S. Relationship between travel distance and utilization of breast cancer treatment in rural northern michigan. JAMA. 2002;287(1):111. doi:10.1001/jama.287.1.111.
Celaya M, Rees J, Gibson J, Riddle B, Greenberg E. Travel distance and season of diagnosis affect treatment choices for women with early-stage breast cancer in a predominantly rural population (United States). Cancer Causes Control. 2006;17(6):851–6.
Ahamad A. Geographic access to cancer care: a disparity and a solution. Postgrad Med J. 2011;87(1031):585–9. doi:10.1136/pgmj.2010.111930.
Wan Y. Examine the effect of geographic distance on breast cancer patients’ utilization of high volume hospitals [Master of Science Thesis]. Iowa. University of Iowa; 2009. http://ir.uiowa.edu/etd/446.
Wee C, McCarthy E, Davis R, Phillips R. Screening for cervical and breast cancer: is obesity an unrecognized barrier to preventive care? Ann Intern Med. 2000;132(9):697–704.
Mitchell KJ, Fritschi L, Reid A, McEvoy SP, Ingram DM, Jamrozik K, et al. Rural-urban differences in the presentation, management and survival of breast cancer in Western Australia. Breast. 2006;15(6):769–76. doi:10.1016/j.breast.2006.04.001.
Gulliford M, Figueroa-Munoz J, Morgan M, Hughes D, Gibson B, Beech R, et al. What does ‘access to health care’ mean? J Health Serv Res Policy. 2002;7(3):186–8. doi:10.1258/135581902760082517.
OECD. Regions at a glance 2007. Regional disparities in GDP per capita. 2007. p. 58–3. Available from: www.sourceoecd.org/governance/9789264009875.
Charalampopoulou A, Petridou E, Spyridopoulos T, Dessypris N, Oikonomou A, Athanasiadou-Piperopoulou F, et al. An integrated evaluation of socioeconomic and clinical factors in the survival from childhood acute lymphoblastic leukaemia: a study in Greece. Eur J Cancer Prev. 2004;13(5):397–401.
Lightfoot TJ, Johnston WT, Simpson J, Smith AG, Ansell P, Crouch S, et al. Survival from childhood acute lymphoblastic leukaemia: the impact of social inequality in the United Kingdom. Eur J Cancer. 2012;48(2):263–9.
Vona-Davis L, Rose DP. The influence of socioeconomic disparities on breast cancer tumor biology and prognosis: a review. J Womens Health (Larchmt). 2009;18(6):883–93. doi:10.1089/jwh.2008.1127.
Loi S, Milne RL, Friedlander ML, McCredie MRE, Giles GG, Hopper JL, et al. Obesity and outcomes in premenopausal and postmenopausal breast cancer. Cancer Epidemiol Biomarkers Prev. 2005;14(7):1686–91. doi:10.1158/1055-9965.epi-05-0042.
Borugian MJ, Sheps SB, Kim-Sing C, Olivotto IA, Van Patten C, Dunn BP, et al. Waist-to-hip ratio and breast cancer mortality. Am J Epidemiol. 2003;158(10):963–8. doi:10.1093/aje/kwg236.
Petrelli JM, Calle EE, Rodriguez C, Thun MJ. Body mass index, height, and postmenopausal breast cancer mortality in a prospective cohort of US women. Cancer Causes Control. 2002;13(4):325–32.
Hall HI, Coates RJ, Uhler RJ, Brinton LA, Gammon MD, Brogan D, et al. Stage of breast cancer in relation to body mass index and bra cup size. Int J Cancer. 1999;82(1):23–7.
Griggs JJ, Sorbero MES, Lyman GH. Undertreatment of obese women receiving breast cancer chemotherapy. Arch Intern Med. 2005;165(11):1267–73. doi:10.1001/archinte.165.11.1267.
McTiernan A, Rajan KB, Tworoger SS, Irwin M, Bernstein L, Baumgartner R, et al. Adiposity and sex hormones in postmenopausal breast cancer survivors. J Clin Oncol. 2003;21(10):1961–6.
Maehle B, Tretli S. Pre-morbid body-mass-index in breast cancer: reversed effect on survival in hormone receptor negative patients. Breast Cancer Res Treat. 1996;41(2):123–30.
Sparano J, Wang M, Martino S, Jones V, Perez E, Saphner T, et al. editors [S2-1]. Obesity at Diagnosis Is Associated with Inferior Outcomes in Hormone Receptor Positive Breast Cancer. 33rd Annual San Antonio Breast Cancer Symposium; 2010; San Antonio, Texas, USA.
Ademuyiwa FO, Groman A, O’Connor T, Ambrosone C, Watroba N, Edge SB. Impact of body mass index on clinical outcomes in triple-negative breast cancer. Cancer. 2011;117(18):4132–40.
Parolin V, Fiorio E, Mercanti A, Riolfi M, Cetto GL, Surmacz E, et al. Impact of BMI on clinical outcome of HER2-positive breast cancer. J Clin Oncol. 2010;28:15s(suppl;abstr 1130).
Goodwin PJ. Insulin in the adjuvant breast cancer setting: a novel therapeutic target for lifestyle and pharmacologic interventions? J Clin Oncol. 2008;26(6):833–4. doi:10.1200/jco.2007.14.7132.
Gunter MJ, Hoover DR, Yu H, Wassertheil-Smoller S, Rohan TE, Manson JE, et al. Insulin, insulin-like growth factor-i, and risk of breast cancer in postmenopausal women. J Natl Cancer Inst. 2009;101(1):48–60. doi:10.1093/jnci/djn415.
Kentikelenis A, Karanikolos M, Papanicolas I, Basu S, McKee M, Stuckler D. Health effects of financial crisis: omens of a Greek tragedy. Lancet. 2011;378(9801):1457–8.
Ethical standards
The present study complies with the current laws of the Greece.
Conflict of interest
All authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix: Clinical studies
Appendix: Clinical studies
-
1.
HE 10/97 trial was a randomized phase III trial in patients with high-risk node-negative or node-positive operable breast cancer, comparing four cycles of epirubicin followed by four cycles of CMF (E-CMF) with three cycles of epirubicin, followed by three cycles of paclitaxel followed by three cycles of CMF (E-T-CMF). All cycles were given every two weeks with granulocyte-colony stimulating factor (G-CSF) support. Dose intensity of all drugs in both treatment arms were identical, but cumulative doses and duration of chemotherapy period differed. Totally, 595 eligible patients entered the study in a period of 3.5 years (1997–2000).
-
2.
HE 10/00 was a randomized phase III trial in which a total of 1086 eligible patients with node-positive operable breast cancer were accrued in a period of 5 years (2000–2005). Patients were treated with E-T-CMF (exactly in the HE 10/97 trial) or with four cycles of epirubicin/paclitaxel combination every three weeks followed by three cycles of CMF every two weeks (ET-CMF). By study design, the cumulative doses and the duration of chemotherapy period were identical in the two arms but dose–intensity of epirubicin and paclitaxel was double in the E-T-CMF arm. Treatment schedules in the two studies are described in detail in Table S1 (Supplementary only). Baseline characteristics and clinical outcomes have been published.
-
3.
HE 10/04 were two consecutively conducted feasibility studies in which 44 and 45 eligible patients with node-positive or high-risk node-negative breast cancer were accrued in 2004. Patients were treated with three cycles of epirubicin followed by three cycles of CMF (at the same dosage as in the HE 10/97 trial) followed by nine weekly cycles of paclitaxel or nine weekly cycles of docetaxel respectively.
-
4.
HE 10/05 was a randomized phase III trial in which a total 1001 eligible patients with node-positive or high-risk node-negative operable breast cancer were accrued in a period of 3 years (2005–2008). Patients were treated with E-T-CMF (as in the 10/97 trial but with a lower dose of paclitaxel, 200 mg/m2) or 3 cycles of epirubicin followed by 3 cycles of intensified CMF followed by 8 cycles of weekly paclitaxel or docetaxel (as in HE 10/04 trial) (unpublished data). All studies were designed and conducted by the Hellenic Cooperative Oncology Group (HeCOG).
All clinical protocols were approved by local regulatory authorities. Additionally, the randomized trials HE 10/97, HE 10/00 and HE 10/05 were included in the Australian New Zealand Clinical Trials Registry (ANZCTR) and allocated the following Registration Numbers: ACTRN12611000506998, ACTRN12609001036202 and ACTRN12610000151033, respectively. Each patient signed a study-specific written informed consent before randomization which in addition allowed the use of biological material for future research purposes.
Rights and permissions
About this article
Cite this article
Panagopoulou, P., Gogas, H., Dessypris, N. et al. Survival from breast cancer in relation to access to tertiary healthcare, body mass index, tumor characteristics and treatment: a Hellenic Cooperative Oncology Group (HeCOG) study. Eur J Epidemiol 27, 857–866 (2012). https://doi.org/10.1007/s10654-012-9737-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10654-012-9737-z