Abstract
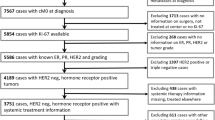
In a neoadjuvant setting, three parameters for Ki-67 could be obtained: pre-treatment Ki-67, post-treatment Ki-67 and Ki-67 change between pre- and post-treatments. It is uncertain which of the three parameters has the greatest prognostic significance, and whether this parameter has significance in each subtype of breast cancer. A total of 385 patients who received neoadjuvant anthracycline followed by taxane chemotherapy and subsequent surgery for breast cancer were analyzed retrospectively. By immunohistochemistry (IHC), patients were divided into four subtypes (Luminal A, Luminal B, Triple negative, and HER2). Ki-67 was examined by IHC in pre-treatment core needle samples and post-treatment surgical excision specimens. The relapse-free survival (RFS) rate was compared among each subtype. The median follow-up period was 56 months. The rate of pathological complete response was higher for HER2 (34.8 %) and Triple negative (24.3 %) subtypes than for Luminal B (8.3 %) and Luminal A (3.8 %) subtypes (p < 0.0001). A reduction in Ki-67 was observed in 58.5, 83.4, 70.2, and 74.2 % of patients in the Luminal A, Luminal B, Triple negative, and HER2 subtypes, respectively. Ki-67 change between pre- and post-treatments was an independent prognostic factor, but pre-treatment Ki-67 and post-treatment Ki-67 were not independent prognostic factors in a multivariate analysis. The RFS was significantly different between patients whose Ki-67 was reduced and those not reduced for Luminal B (81.4 vs. 50.0 %, p = 0.006), Triple negative (74.8 vs. 43.5 %, p = 0.006) and HER2 (82.7 vs. 59.0 %, p = 0.009). However, for Luminal A, the difference in RFS was not associated with changes of Ki-67 (78.8 vs. 75.3 %, p = 0.193). Ki-67 change between pre- and post-neoadjuvant chemotherapy is an independent prognostic factor in patients of Luminal B, Triple negative, and HER2 subtypes. Pre-treatment Ki-67 and post-treatment Ki-67 were not independent prognostic factors in a multivariate analysis.







Similar content being viewed by others
References
Kaufmann M, von Minckwitz G, Bear HD, Buzdar A, McGale P, Bonnefoi H, Colleoni M, Denkert C, Eiermann W, Jackesz R, Makris A, Miller W, Pierga JY, Semiglazov V, Schneeweiss A, Souchon R, Stearns V, Untch M, Loibl S (2007) Recommendations from an international expert panel on the use of neoadjuvant (primary) systemic treatment of operable breast cancer: new perspectives 2006. Annal Oncol 18(12):1927–1934. doi:10.1093/annonc/mdm201
Bonadonna G, Valagussa P (1996) Primary chemotherapy in operable breast cancer. Semin Oncol 23(4):464–474
Ellis PA, Smith IE (1996) Primary chemotherapy for early breast cancer. Cancer Treat Rev 22(6):437–450
Bonadonna G, Valagussa P, Brambilla C, Ferrari L, Moliterni A, Terenziani M, Zambetti M (1998) Primary chemotherapy in operable breast cancer: eight-year experience at the Milan Cancer Institute. J Clin Oncol 16(1):93–100
Kaufmann M, Hortobagyi GN, Goldhirsch A, Scholl S, Makris A, Valagussa P, Blohmer JU, Eiermann W, Jackesz R, Jonat W, Lebeau A, Loibl S, Miller W, Seeber S, Semiglazov V, Smith R, Souchon R, Stearns V, Untch M, von Minckwitz G (2006) Recommendations from an international expert panel on the use of neoadjuvant (primary) systemic treatment of operable breast cancer: an update. J Clin Oncol 24(12):1940–1949. doi:10.1200/JCO.2005.02.6187
van der Hage JA, van de Velde CJ, Julien JP, Tubiana-Hulin M, Vandervelden C, Duchateau L (2001) Preoperative chemotherapy in primary operable breast cancer: results from the European Organization for Research and Treatment of Cancer trial 10902. J Clin Oncol 19(22):4224–4237
Fisher B, Bryant J, Wolmark N, Mamounas E, Brown A, Fisher ER, Wickerham DL, Begovic M, DeCillis A, Robidoux A, Margolese RG, Cruz AB Jr, Hoehn JL, Lees AW, Dimitrov NV, Bear HD (1998) Effect of preoperative chemotherapy on the outcome of women with operable breast cancer. J Clin Oncol 16(8):2672–2685
Gerdes J, Li L, Schlueter C, Duchrow M, Wohlenberg C, Gerlach C, Stahmer I, Kloth S, Brandt E, Flad HD (1991) Immunobiochemical and molecular biologic characterization of the cell proliferation-associated nuclear antigen that is defined by monoclonal antibody Ki-67. Am J Pathol 138(4):867–873
Gerdes J, Lemke H, Baisch H, Wacker HH, Schwab U, Stein H (1984) Cell cycle analysis of a cell proliferation-associated human nuclear antigen defined by the monoclonal antibody Ki-67. J Immunol 133(4):1710–1715
Urruticoechea A, Smith IE, Dowsett M (2005) Proliferation marker Ki-67 in early breast cancer. J Clin Oncol 23(28):7212–7220. doi:10.1200/JCO.2005.07.501
Colozza M, Azambuja E, Cardoso F, Sotiriou C, Larsimont D, Piccart MJ (2005) Proliferative markers as prognostic and predictive tools in early breast cancer: where are we now? Ann Oncol 16(11):1723–1739. doi:10.1093/annonc/mdi352
de Azambuja E, Cardoso F, de Castro G Jr, Colozza M, Mano MS, Durbecq V, Sotiriou C, Larsimont D, Piccart-Gebhart MJ, Paesmans M (2007) Ki-67 as prognostic marker in early breast cancer: a meta-analysis of published studies involving 12,155 patients. Br J Cancer 96(10):1504–1513. doi:10.1038/sj.bjc.6603756
Goldhirsch A, Wood WC, Coates AS, Gelber RD, Thurlimann B, Senn HJ (2011) Strategies for subtypes–dealing with the diversity of breast cancer: highlights of the St. Gallen international expert consensus on the primary therapy of early breast cancer 2011. Annal Oncol 22(8):1736–1747. doi:10.1093/annonc/mdr304
Keam B, Im SA, Kim HJ, Oh DY, Kim JH, Lee SH, Chie EK, Han W, Kim DW, Moon WK, Kim TY, Park IA, Noh DY, Heo DS, Ha SW, Bang YJ (2007) Prognostic impact of clinicopathologic parameters in stage II/III breast cancer treated with neoadjuvant docetaxel and doxorubicin chemotherapy: paradoxical features of the triple negative breast cancer. BMC Cancer 7:203. doi:10.1186/1471-2407-7-203
Archer CD, Parton M, Smith IE, Ellis PA, Salter J, Ashley S, Gui G, Sacks N, Ebbs SR, Allum W, Nasiri N, Dowsett M (2003) Early changes in apoptosis and proliferation following primary chemotherapy for breast cancer. Br J Cancer 89(6):1035–1041. doi:10.1038/sj.bjc.6601173
Jones RL, Salter J, A’Hern R, Nerurkar A, Parton M, Reis-Filho JS, Smith IE, Dowsett M (2009) The prognostic significance of Ki-67 before and after neoadjuvant chemotherapy in breast cancer. Breast Cancer Res Treat 116(1):53–68. doi:10.1007/s10549-008-0081-7
Dowsett M, Smith IE, Ebbs SR, Dixon JM, Skene A, A’Hern R, Salter J, Detre S, Hills M, Walsh G (2007) Prognostic value of Ki-67 expression after short-term presurgical endocrine therapy for primary breast cancer. J Natl Cancer Inst 99(2):167–170. doi:10.1093/jnci/djk020
Matsubara N, Mukai H, Itoh K, Nagai S (2011) Prognostic impact of Ki-67 overexpression in subgroups categorized according to St. Gallen with early stage breast cancer. Oncology 81(5–6):345–352. doi:10.1159/000334920
Klintman M, Bendahl PO, Grabau D, Lovgren K, Malmstrom P, Ferno M (2010) The prognostic value of Ki-67 is dependent on estrogen receptor status and histological grade in premenopausal patients with node-negative breast cancer. Mod Pathol 23(2):251–259. doi:10.1038/modpathol.2009.167
Lee J, Im YH, Lee SH, Cho EY, Choi YL, Ko YH, Kim JH, Nam SJ, Kim HJ, Ahn JS, Park YS, Lim HY, Han BK, Yang JH (2008) Evaluation of ER and Ki-67 proliferation index as prognostic factors for survival following neoadjuvant chemotherapy with doxorubicin/docetaxel for locally advanced breast cancer. Cancer Chemother Pharmacol 61(4):569–577. doi:10.1007/s00280-007-0506-8
Bottini A, Berruti A, Bersiga A, Brizzi MP, Bruzzi P, Aguggini S, Brunelli A, Bolsi G, Allevi G, Generali D, Betri E, Bertoli G, Alquati P, Dogliotti L (2001) Relationship between tumour shrinkage and reduction in Ki-67 expression after primary chemotherapy in human breast cancer. Br J Cancer 85(8):1106–1112. doi:10.1054/bjoc.2001.2048
Faneyte IF, Schrama JG, Peterse JL, Remijnse PL, Rodenhuis S, van de Vijver MJ (2003) Breast cancer response to neoadjuvant chemotherapy: predictive markers and relation with outcome. Br J Cancer 88(3):406–412. doi:10.1038/sj.bjc.6600749
Colleoni M, Viale G, Zahrieh D, Pruneri G, Gentilini O, Veronesi P, Gelber RD, Curigliano G, Torrisi R, Luini A, Intra M, Galimberti V, Renne G, Nole F, Peruzzotti G, Goldhirsch A (2004) Chemotherapy is more effective in patients with breast cancer not expressing steroid hormone receptors: a study of preoperative treatment. Clin Cancer Res 10(19):6622–6628. doi:10.1158/1078-0432.CCR-04-0380
Billgren AM, Rutqvist LE, Tani E, Wilking N, Fornander T, Skoog L (1999) Proliferating fraction during neoadjuvant chemotherapy of primary breast cancer in relation to objective local response and relapse-free survival. Acta Oncol 38(5):597–601
Takada M, Kataoka A, Toi M, Bando H, Toyama K, Horiguchi S, Ueno T, Linder S, Saji S, Hayashi Y, Funata N, Kinoshita J, Murakami S, Ohono S (2004) A close association between alteration in growth kinetics by neoadjuvant chemotherapy and survival outcome in primary breast cancer. Int J Oncol 25(2):397–405
von Minckwitz G, Untch M, Blohmer JU, Costa SD, Eidtmann H, Fasching PA, Gerber B, Eiermann W, Hilfrich J, Huober J, Jackisch C, Kaufmann M, Konecny GE, Denkert C, Nekljudova V, Mehta K, Loibl S (2012) Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol 30(15):1796–1804. doi:10.1200/JCO.2011.38.8595
Acknowledgments
The study was carried out in accordance with the Declaration of Helsinki and Japanese ethical guidelines for epidemiological research.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matsubara, N., Mukai, H., Fujii, S. et al. Different prognostic significance of Ki-67 change between pre- and post-neoadjuvant chemotherapy in various subtypes of breast cancer. Breast Cancer Res Treat 137, 203–212 (2013). https://doi.org/10.1007/s10549-012-2344-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-2344-6