Abstract
Background
This multi-center, phase III trial assesses the efficacy of daikenchuto (TU-100) on gastrointestinal disorders after hepatic resection (UMIN Registration No. 000003103).
Materials and methods
A total of 231 patients, who underwent hepatic resection at 26 Japanese centers, were enrolled. Patients were randomly assigned to receive either oral doses (15 g/day, three times a day) of TU-100 or placebo control from preoperative day 3 to postoperative day 10, except on the day of surgery. Primary end points were the time from extubation until the first postoperative bowel movement (FBM-T), serum C-reactive protein (CRP) and ammonia levels.
Results
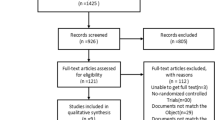
Finally, 209 patients (TU-100: n = 108, placebo: n = 101) were included in the statistical analysis. The median FBM-T was 88.2 h (95 % CI 74.0–94.1) in the TU-100 group and 93.1 h (95 % CI 83.3–99.4) in the placebo group, demonstrating that TU-100 accelerated the time to first bowel movement significantly more than placebo control. Serum CRP levels did not differ significantly during the study period, although serum CRP levels in the TU-100 group tended to be lower than those in the placebo group in patients with grade B liver damage. Meanwhile, the two groups had similar serum ammonia levels. TU-100-related serious adverse events did not occur during the study.
Conclusions
TU-100 appears to improve gastrointestinal dysmotility and reduce serum CRP levels in patients with grade B liver damage after hepatectomy. TU-100 is an effective treatment option after hepatic resection in patients with liver cancer.



Similar content being viewed by others
Abbreviations
- TU-100:
-
Daikenchuto
- JFMC:
-
Japanese Foundation for Multidisciplinary Treatment of Cancer
- FBM-T:
-
The time from extubation until the first postoperative bowel movement
- CRP:
-
C-reactive protein
- COX-2:
-
Cyclooxygenase-2
- CGRP:
-
Calcitonin gene-related peptide
- VIP:
-
Vasoactive intestinal polypeptide
- AUC:
-
Area under curve
- SD:
-
Standard deviation
- CRLR:
-
Calcitonin receptor like receptor
- Ramp1:
-
Receptor activity modifying protein 1
- ADM:
-
Adrenomedullin
References
Furukawa Y, Hanyu N, Nakada K et al (1999) Effects of the Chinese herb medicine “Dai-kenchu-to” on GI motility and for treatment of bowel obstruction. Gastroenterology 116:A811
Iwai N, Kume Y, Kimura O et al (2007) Effects of herbal medicine Dai-Kenchu-to on anorectal function in children with severe constipation. Eur J Pediatr Surg 17:115–118
Sakakibara R, Odaka T, Lui Z et al (2005) Dietary herb extract dai-kenchu-to ameliorates constipation in parkinsonian patients (Parkinson’s disease and multiple system atrophy). Mov Disord 20:261–262
Ohya T, Usui Y, Arii S et al (2003) Effect of dai-kenchu-to on obstructive bowel disease in children. Am J Chin Med 31:129–135
Nakamura T, Sakai A, Isogami I et al (2002) Abatement of morphine-induced slowing in gastrointestinal transit by Dai-kenchu-to, a traditional Japanese herbal medicine. Jpn J Pharmacol 88:217–221
Satoh K, Kase Y, Yuzurihara M et al (2003) Effect of Dai-kenchu-to (Da-Jian-Zhong-Tang) on the delayed intestinal propulsion induced by chlorpromazine in mice. J Ethnopharmacol 86:37–44
Satoh K, Hayakawa T, Kase Y et al (2001) Mechanisms for contractile effect of dai-kenchu-to in isolated guinea pig ileum. Dig Dis Sci 46:250–256
Hayakawa T, Kase Y, Saito K et al (1999) Effects of Dai-kenchu-to on intestinal obstruction following laparotomy. J Smooth Muscle Res 35:47–54
Yoshikawa K, Kurita N, Higashijima J et al (2008) Kampo medicine “Dai-kenchu-to” prevents bacterial translocation in rats. Dig Dis Sci 53:1824–1831
Chikakiyo M, Shimada M, Nakao T et al (2012) Kampo medicine “Dai-kenchu-to” prevents CPT-11-induced small-intestinal injury in rats. Surg Today 42:60–67
Sato Y, Katagiri F, Inoue S et al (2004) Dai-kenchu-to raises levels of calcitonin gene-related peptide and substance P in human plasma. Biol Pharm Bull 27:1875–1877
Sato Y, Inoue S, Katagiri F et al (2006) Effects of pirenzepine on Dai-kenchu-to-induced elevation of the plasma neuropeptide levels in humans. Biol Pharm Bull 29:166–171
Suehiro T, Matsumata T, Shikada Y et al (2005) The effect of the herbal medicines dai-kenchu-to and keishi-bukuryo-gan on bowel movement after colorectal surgery. Hepatogastroenterology 52:97–100
Endo S, Nishida T, Nishikawa K et al (2006) Dai-kenchu-to, a Chinese herbal medicine, improves stasis of patients with total gastrectomy and jejunal pouch interposition. Am J Surg 192:9–13
Kaiho T, Tanaka T, Tsuchiya S et al (2005) Effect of the herbal medicine Dai-kenchu-to on serum ammonia in hepatectomized patients. Hepatogastroenterology 52:161–165
Kono T, Kanematsu T, Kitajima M (2009) Exodus of Kampo, traditional Japanese medicine, from the complementary and alternative medicines: is it time yet? Surgery 146:837–840
Manabe N, Camilleri M, Rao A et al (2010) Effect of daikenchuto (TU-100) on gastrointestinal and colonic transit in humans. Am J Physiol Gastrointest Liver Physiol 298:G970–G975
Liver Cancer Study Group of Japan (1997) Classification of primary liver cancer, 1 English edn. Kanehara, Tokyo
Nagano T, Itoh H, Takeyama M (2000) Effects of Dai-kenchu-to on levels of 5-hydroxytryptamine (serotonin) and vasoactive intestinal peptides in human plasma. Biol Pharm Bull 23:352–353
Nagano T, Itoh H, Takeyama M (1999) Effect of Dai-kenchu-to on levels of 3 brain-gut peptides (motilin, gastrin and somatostatin) in human plasma. Biol Pharm Bull 22:1131–1133
Itoh T, Yamakawa J, Mai M et al (2002) The effect of the herbal medicine daikenchu-to on post-operative ileus. J Int Med Res 30:428–432
Yoshikawa K, Shimada M, Nishioka M et al (2012) The effects of the Kampo medicine (Japanese herbal medicine) “Daikenchuto” on the surgical inflammatory response following laparoscopic colorectal resection. Surg Today 42:646–651
Kito Y, Suzuki H (2006) Effects of Dai-kenchu-to on spontaneous activity in the mouse small intestine. J Smooth Muscle Res 42:189–201
Fukuda H, Chen C, Mantyh C et al (2006) The herbal medicine, Dai-Kenchu-to, accelerates delayed gastrointestinal transit after an operation on rats. J Surg Res 131:290–295
Tokita Y, Yuzurihara M, Sakaguchi M et al (2007) The pharmacological effects of Daikenchuto, a traditional herbal medicine, on delayed gastrointestinal transit in rat postoperative ileus. J Pharmacol Sci 104:303–310
Satoh K, Kase Y, Hayakawa T et al (2001) Dai-kenchu-to enhances accelerated small intestinal movement. Biol Pharm Bull 24:1122–1126
Kawasaki N, Nakada K, Nakayoshi T et al (2007) Effect of Dai-kenchu-to on gastrointestinal motility based on differences in the site and timing of administration. Dig Dis Sci 52:2684–2694
Shibata C, Sasaki I, Naito H et al (1999) The herbal medicine Dai-Kenchu-Tou stimulates upper gut motility through cholinergic and 5-hydroxytryptamine 3 receptors in conscious dogs. Surgery 126:918–924
Kinoshita Y, Inui T, Chiba T (1993) Calcitonin gene-related peptide: a neurotransmitter involved in capsaicin-sensitive afferent nerve-mediated gastric mucosal protection. J Clin Gastroenterol 17:S27–S32
Reinshagen M, Patel A, Sottili M et al (1994) Protective function of extrinsic sensory neurons in acute rabbit experimental colitis. Gastroenterology 106:1208–1214
Kono T, Koseki T, Chiba S et al (2008) Colonic vascular conductance increased by Daikenchuto via calcitonin gene-related peptide and receptor-activity modifying protein 1. J Surg Res 150:78–84
Genesca J, Gonzalez A, Catalan R et al (1999) Adrenomedullin, a vasodilator peptide implicated in hemodynamic alterations of liver cirrhosis: relationship to nitric oxide. Dig Dis Sci 44:372–376
Kono T, Kaneko A, Hira Y et al (2010) Anti-colitis and -adhesion effects of daikenchuto via endogenous adrenomedullin enhancement in Crohn’s disease mouse model. J Crohns Colitis 4:161–170
Kono T, Omiya Y, Hira Y et al (2011) Daikenchuto (TU-100) ameliorates colon microvascular dysfunction via endogenous adrenomedullin in Crohn’s disease rat model. J Gastroenterol 46:1187–1196
Ogasawara T, Morine Y, Ikemoto T et al (2008) Influence of Dai-kenchu-to (DKT) on human portal blood flow. Hepatogastroenterology 55:574–577
Narita M, Hatano E, Tamaki N et al (2009) Dai-kenchu-to attenuates rat sinusoidal obstruction syndrome by inhibiting the accumulation of neutrophils in the liver. J Gastroenterol Hepatol 24:1051–1057
Seo YS, Shah VH (2012) The role of gut-liver axis in the pathogenesis of liver cirrhosis and portal hypertension. Clin Mol Hepatol 18:337–346
Bellot P, Francés R, Such J (2013) Pathological bacterial translocation in cirrhosis: pathophysiology, diagnosis and clinical implications. Liver Int 33:31–39
Basu S (2003) Carbon tetrachloride-induced lipid peroxidation: eicosanoid formation and their regulation by antioxidant nutrients. Toxicology 189:113–127
Acknowledgments
This work was jointly supported by the Japanese Foundation for Multidisciplinary Treatment of Cancer.
Conflict of interest
Mitsuo Shimada received the research grant by TSUMURA & CO. (Tokyo, Japan). The other authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
For the JFMC40-1001 group.
Appendix
Appendix
The members of this multicenter, phase III trial (JFMC40-1001) group were as follows:
Steering Committee: Mitsuo Kusano, Junichi Sakamoto, Mitsuo Shimada, Toru Kono, Takashi Kaiho, Toshiya Kamiyama, Hiroaki Nagano, Minoru Tanabe, Yuji Morine;
Medical Advisors: Masaki Kitajima, Takashi Kanematsu, Shigetoyo Saji;
Independent Data and Safety Monitoring Committee: Masaru Miyazaki, Kazuhiro Hanasaki;
Statistical Analyst: Satoshi Morita.
Principal Investigators (all in Japan): Hokkaido University Hospita—T. Kamiyama, K. Nakanishi; Supporo Medical University—K. Hirata, T. Mizuguchi; Kushiro Rosai Hospital—K. Ogasawara; Hitachi General Hospital—M. Okumura, K. Ueda; Fukaya Red Cross Hospital—H. Ito; Kameda General Hospital—N. Kano, S. Yamada; Kimitsu Chuo Hospital—T. Kaiho; Toho University Omori Medical Center—H. Kaneko, Y. Otsuka; Tokyo Medical University Hachioji Medical Center—M. Shimazu, Y. Abe; Toyama University Hospital—K. Tsukada, K. Matsui; Fujita Health University—A. Sugioka, T. Tokoro; Fujita Health University School of Medicine Banbuntane Hotokukai Hospital—Z. Morise; Kyoto University Hospital—E. Hatano, K. Taura; Osaka Medical Center for Cancer and Cardiovascular Diseases—T. Yamada, K. Goto; Osaka City University Hospital—S. Kubo, T. Uenishi; Osaka General Medical Center—K. Nishikawa, T. Deguchi; Osaka University—H. Nagano, S. Kobayashi; Osaka Medical College—K. Uchiyama, M. Hayashi; National Hospital Organization Osaka Minami Medical Center—K. Tabuse, Y. Shono; Tokushima University Hospital—M. Shimada, Y. Morine; Kyusyu University Hospital—Y. Maehara, K. Shirabe; Iizuka Hospital—K. Kajiyama, N. Harimoto; Nagasaki University Graduate School of Biomedical Science—S. Eguchi, M. Takatsuki; Kumamoto University Hospital—H. Baba, T. Beppu; Oita University Hospital—S. Kitano, M. Ota; Kagoshima University Hospital—S. Natsugoe, S. Ueno.
About this article
Cite this article
Shimada, M., Morine, Y., Nagano, H. et al. Effect of TU-100, a traditional Japanese medicine, administered after hepatic resection in patients with liver cancer: a multi-center, phase III trial (JFMC40-1001). Int J Clin Oncol 20, 95–104 (2015). https://doi.org/10.1007/s10147-014-0678-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-014-0678-2