Abstract
Purpose
Hand–foot syndrome (HFS) is a common adverse event that can be induced by capecitabine. It is hypothesized that capecitabine (Hoffmann-La Roche Inc.) based chemotherapy can cause overexpression of COX-2 in tumor and healthy tissue, which finally induced HFS in hands and feet. Based on this, we believed that a selected COX-2 inhibitor (celecoxib, Pfizer Pharmaceuticals LLC) could ease HFS. We designed a prospective clinical study to test the hypothesis.
Methods
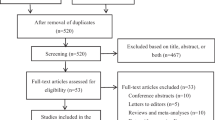
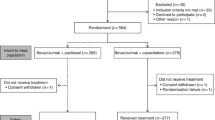
From August 2008 to January 2010, 110 patients with stage II/III colorectal cancer who were eligible for adjuvant chemotherapy were enrolled in the study and divided into 4 groups by random, but 9 patients did not finish at least 4 cycles of chemotherapy. There were sixteen patients in capecitabine group, and fifteen patients in capecitabine and celecoxib group. Thirty-four patients were in XELOX (capecitabine plus oxaliplatine) group, and thirty-six patients in XELOX+ celecoxib group. All 101 patients finished chemotherapy and follow-up interviews.
Results
The group that had received capecitabine and celecoxib had a significantly reduced frequency of >grade 1 hand–foot syndrome (29 vs. 72% P < 0.001), and >grade 2 (11.76% vs. 30% P = 0.024). Five patients experienced grade 3 HFS in capecitabine group and only 1 patient had grade 3 HFS in capecitabine and celecoxib group. There were 5 patients in capecitabine group who refused to go on chemotherapy because of HFS, but there was none in capecitabine and celecoxib group.
Conclusions
From the result of this study, we could learn that celecoxib could reduce HFS that was induced by capecitabine. So we recommend that celecoxib can be used in capecitabine-based chemotherapy.
Similar content being viewed by others
References
Baack BR, Burgdorf WH (1991) Chemotherapy-induced acral erythema. J Am Acad Dermatol 24:457–461
Cassidy J, Tabernero J, Twelves C et al (2004) XELOX (capecitabine plus oxaliplatin): active firstline therapy for patients with metastatic colorectal cancer. J Clin Oncol 22:2084–2091
Cianchi F, Cortesini C, Bechi P, Fantappiè O, Messerini L, Vannacci A, Sardi I, Baroni G, Boddi V, Mazzanti R, Masini E (2001) Up-regulation of cyclooxygenase 2 gene expression correlates with tumor angiogenesis in human colorectal cancer. Gastroenterology 121:1339–1347
Dong ZW, Qiao YL, Li LD, Chen YD, Wang RT, Lei TH et al (2002) Report of Chinese cancer control strategy. Chin Cancer 11:250–260
Edward LIN, Jeffrey S, Morris et al (2002) Effect of celecoxib on capecitabine-induced hand-foot syndrome and antitumor activity. Oncology (Williston Park) 16:31–37
Edward LIN, Steven A, Curley, Christopher C, Crane et al (2006) Retrospective study of capecitabine and celecoxib in metastatic colorectal cancer: potential benefits and COX-2 as the common mediator in pain, toxicities and survival? Am J Clin Oncol 29:232–239
Gill S, Loprinzi CL, Sargent DJ et al (2004) Pooled analysis of fluorouracil-based adjuvant therapy for stage II and III colon cancer: who benefits and by how much? J Clin Oncol 22:1797–1806
Gressett SM, Stanford BL, Hardwicke F (2002) Management of hand-foot syndrome induced by capecitabine. J Oncol Pharm Pract 12:131–141
Gupta RA, DuBois RN (2001) Colorectal cancer prevention and treatment by inhibition of cyclooxygenase. Nat Rev Cancer 1:11–21
Howe LR, Dannenberg AJ (2002) A role for cyclooxygenase-2 inhibitors in the prevention and treatment of cancer. Semin Oncol 29:111–119
Jemal A, Siegel R, Ward E et al (2008) Cancer statistics. CA Cancer J Clin 58:71–96
Moertel CG, Fleming TR, Macdonald JS et al (1990) Levamisole and fluorouracil for adjuvant therapy of resected colon carcinoma. New Engl J Med 32:352–358
Nagore E, Insa A, Sanmartin O (2000) Antineoplastic therapy-induced palmar plantar erythrodysesthesia (‘hand-foot’) syndrome: incidence, recognition and management. Am J Clin Dermatol 1:225–234
Schmoll H-J, Cartwright T, Tabernero J et al (2007) Phase III trial of capecitabine plus oxaliplatin as adjuvant therapy for stage III colon cancer: a planned safety analysis in 1, 864 Patients. J Clin Oncol 25:102–108
Sheehan KM, Sheahan K, O’Donoghue DP, MacSweeney F, Conroy RM, Fitzgerald DJ, Murray FE (1999) The relationship between cyclooxygenase-2 expression and colorectal cancer. JAMA 282:1254–1257
Steinbach G, Lynch PM, Phillips RK, Wallace MH, Hawk E, Gordon GB, Wakabayashi N, Saunders B, Shen Y, Fujimura T, Su LK, Levin B (2000) The effect of celecoxib, a cyclooxygenase-2 inhibitor, in familial adenomatous polyposis. N Engl J Med 342:1946–1952
Twelves C, Wong A, Nowacki MP, Abt M, Burris H 3rd, Carrato A et al (2005) Capecitabine as adjuvant treatment for stage III colon cancer. N Engl J Med 352:2696–2704
Yong J, Wei W, Ping L et al (2007) Controlled clinical research of Celecoxib In prophylaxising side effects caused by Capecitabine. Chinese J Composite Clinical Hygiene 9:6–8
Zuehlke R (1974) Erythematous eruption of the palms and soles associated with mitotane therapy. Dermatologica 148:90–92
Acknowledgments
We are very thankful to Zhi-heng Peng and Zi-yi Huang for the acquisition of data.
Conflict of interest
We declare that there is no conflict of interest in this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Rong-Xin Zhang and Xiao-Jun Wu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhang, RX., Wu, XJ., Lu, SX. et al. The effect of COX-2 inhibitor on capecitabine-induced hand–foot syndrome in patients with stage II/III colorectal cancer: a phase II randomized prospective study. J Cancer Res Clin Oncol 137, 953–957 (2011). https://doi.org/10.1007/s00432-010-0958-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-010-0958-9