Abstract
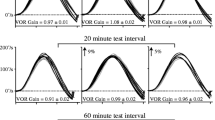
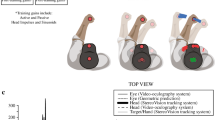
Studies on motor learning typically present a constant adaptation stimulus, corresponding to the desired final adaptive state. Studies of the auditory and optokinetic systems provide compelling evidence that neural plasticity is enhanced when the error signal driving adaptation is instead adjusted gradually throughout training. We sought to determine whether the angular vestibulo-ocular reflex (aVOR) may be adaptively increased using an incremental velocity error signal (IVE) compared with a conventional constant and large velocity-gain demand (x2). We compared the magnitude of aVOR gain change for these two paradigms across different motion contexts (active and passive). Seven individuals with normal vestibular function and six individuals with unilateral vestibular hypofunction (UVH) were exposed to the IVE and x2 (“control”) aVOR demand tasks. Each subject participated in 10 epochs of 30 active head impulses over a 15 min aVOR gain increase training session separately for the IVE and x2 paradigms, separated by either seven days (normal subjects) or 14 days (UVH subjects). For both normal and UVH subjects, both paradigms led to aVOR gain increase during the training session. For the normal subjects, the IVE paradigm led to larger aVOR gain change after training compared to the x2 paradigm, for both active (mean 17.3 ± 4% vs. mean 7.1 ± 9%, P = 0.029) and passive (mean 14.2 ± 5% vs. 4.5 ± 8%, P = 0.018) head impulses. For subjects with UVH, IVE produced a greater change in aVOR gain for active head impulses (mean 18.2 ± 9.2% vs. mean −6 ± 3.8%, P = 0.003). However, aVOR gains for passive head impulses were less consistent after IVE, with only two subjects displaying greater aVOR gain with this incremental paradigm. Some individuals generated compensatory saccades that occurred in the same direction of the deficient aVOR during either training paradigm. Our data suggest that the aVOR is modifiable when the velocity error signal is presented incrementally, and that this adaptation stimulus is particularly effective in the case of unilateral vestibular hypofunction. This has implications for programs of vestibular rehabilitation, where active head rotation is prescribed as a means to improve gaze stability.






Similar content being viewed by others
References
Aw ST, Haslwanter T, Halmagyi GM, Curthoys IS, Yavor RA, Todd MJ (1996) Three-dimensional vector analysis of the human vestibuloocular reflex in response to high-acceleration head rotations. I. Responses in normal subjects. J Neurophysiol 76:4009–4020
Barnes GR, Paige GD (2004) Anticipatory VOR suppression induced by visual and nonvisual stimuli in humans. J Neurophysiol 92:1501–1511
Black RA, Halmagyi GM, Thurtell MJ, Todd MJ, Curthoys IS (2005) The active head-impulse test in unilateral peripheral vestibulopathy. Arch Neurol 62(2):290–293
Bockisch CJ, Straumann D, Hess K, Haslwanter T (2004) Enhanced smooth pursuit eye movements in patients with bilateral vestibular deficits. Neuroreport 15:2617–2620
Bronstein AM, Morland AB, Ruddock KH, Gresty MA (1995) Recovery from bilateral vestibular failure: implications for visual and cervico-ocular function. Acta Otolaryngol Suppl 520(Pt 2):405–407
Burke MR, Barnes GR (2006) Quantitative differences in smooth pursuit and saccadic eye movements. Exp Brain Res 175(4):596–608 Epub 2006 Jul 11
Crane BT, Demer JL (1997) Human gaze stabilization during natural activities: translation, rotation, magnification, target distance effects. J Neurophysiol 78:2129–2144
Dawson-Saunders B, Trapp RG (1994) Basic and clinical biostatistics. Appleton & Lange, Norwalk
Della Santina CC, Cremer PD, Carey JP, Minor LB (2002) Comparison of head thrust test with head autorotation test reveals that the vestibulo-ocular reflex is enhanced during voluntary head movements. Arch Otolaryngol Head Neck Surg 128(9):1044–1054
Eggers SD, De Pennington N, Walker MF, Shelhamer M, Zee DS (2003) Short-term adaptation of the VOR: non-retinal-slip error signals and saccade substitution. Ann N Y Acad Sci Oct 1004:94–110
Gauthier GM, Robinson DA (1975) Adaptation of the human vestibuloocular reflex to magnifying lenses. Brain Res 92(2):331–335
Gonshor A, Melvill Jones G (1976a) Short-term adaptive changes in the human vestibulo-ocular reflex arc. J Physiol (Lond) 256:361–3379
Gonshor A, Melvill Jones G (1976b) Extreme vestibulo-ocular adaptation induced by prolonged optical reversal of vision. J Physiol (Lond) 256:381–414
Grossman GE, Leigh RJ, Abel LA, Lanska DJ, Thurston SE (1988) Frequency and velocity of rotational head perturbations during locomotion. Exp Brain Res 70(3):470–476
Halmagyi GM, Curthoys IS (1998) A clinical sign of canal paresis. Arch Neurol 45(7):737–739
Haslwanter T (1995) Mathematics of three-dimensional eye rotations. Vis Res 35(12):1727–1739
Hepp K (1990) On Listing’s Law. Commun Math Phys 132:285–295
Kagerer FA, Contreras-Vidal JL, Stelmach GE (1997) Adaptation to gradual as compared with sudden visuo-motor distortions. Exp Brain Res 115(3):557–561
Kasai T, Zee DS (1978) Eye-head coordination in labyrinthine-defective human beings. Brain Res 144(1):123–141
Kilgard MP, Merzenich MM (2002) Order-sensitive plasticity in adult primary auditory cortex. Proc Natl Acad Sci USA 99(5):3205–3209
Körding KP, Tenenbaum JB, Shadmehr R (2007) The dynamics of memory as a consequence of optimal adaptation to a changing body. Nat Neurosci 10:779–786
Kowler E, Steinman RM (1979) The effect of expectations on slow oculomotor control. I. Periodic target steps. Vis Res 19:619–632
Migliaccio AA and Todd MJ (1999). Real-time rotation vectors. Aust Phys Eng Sci Med 22:73–80
Nagarajan S, Mahncke H, Salz T, Tallal P, Roberts T, Merzenich MM (1999) Cortical auditory signal processing in poor readers. Proc Natl Acad Sci USA 96(11):6483–6488
Nagarajan SS, Blake DT, Wright BA, Byl N, Merzenich MM (1998) Practice-related improvements in somatosensory interval discrimination are temporally specific but generalize across skin location, hemisphere, and modality. J Neurosci 18(4):1559–1570
Paige GD (1994) Senescence of human visual-vestibular interactions: smooth pursuit, optokinetic, and vestibular control of eye movements with aging. Exp Brain Res 98(2):355–372
Ramat S, Zee DS (2003) Ocular motor responses to abrupt interaural head translation in normal humans. J Neurophysiol 90(2):887–902. Epub 2003 Apr 2
Schubert MC, Das V, Tusa RJ, Herdman SJ (2004) Cervico-ocular reflex in normal subjects and patients with unilateral vestibular hypofunction. Otol Neurotol 25(1):65–71
Schubert MC, Das VE, Tusa RJ, Herdman SJ (2002) Gaze stability during predictable and unpredictable head thrusts. Program No 266.1. Society for Neuroscience. CD ROM, Washington, DC
Schubert MC, Migliaccio AA, Clendaniel RA, Allak A, Carey JP (2008) Mechanism of dynamic visual acuity recovery with vestibular rehabilitation. Arch Phys Med Rehabil 89(3):500–507
Schubert MC, Migliaccio AA, Della Santina CC (2006) Dynamic visual acuity during passive head thrusts in canal planes. J Assoc Res Otolaryngol 7(4):329–338
Shelhamer M, Tiliket C, Roberts D, Kramer PD, Zee DS (1994) Short-term vestibulo-ocular reflex adaptation in humans. II. Error signals. Exp Brain Res 100(2):328–336
Shelhamer M, Ravina B, Kramer PD (1995) Adaptation of the gain of the angular vestibulo-ocular reflex when retinal slip is zero. Soc Neurosci Abstr 21:518
Szturm T, Ireland DJ, Lessing-Turner M (1994) Comparison of different exercise programs in the rehabilitation of patients with chronic peripheral vestibular dysfunction. J Vestib Res 4(6):461–479
Tian J, Crane BT, Demer JL (2000) Vestibular catch-up saccades in labyrinthine deficiency. Exp Brain Res 131(4):448–457
Viirre E, Draper M, Gailey C, Miller D, Furness T (1998) Adaptation of the AVOR in patients with low AVOR gains. J Vestib Res 8(4):331–334
Viirre E, Sitarz R (2002) Vestibular rehabilitation using visual displays: preliminary study. Laryngoscope 112(3):500–503
Yasui S, Young LR (1975) Perceived visual motion as effective stimulus to pursuit eye movement system. Sci 190(4217):906–908
Acknowledgments
We thank Dr. Americo A Migliaccio for assistance with statistical analysis and Dr. Mark Walker and Mr. Dale Roberts for programming expertise. MCS was supported by the National Institute on Deafness and Other Communication Disorders (K23-007926), CDS NIDCD (K08-DC006216, R01-DC009255) and MJS (NSF BCS-0615106 and NIH EB001914). No commercial party has a direct financial interest in the results.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schubert, M.C., Della Santina, C.C. & Shelhamer, M. Incremental angular vestibulo-ocular reflex adaptation to active head rotation. Exp Brain Res 191, 435–446 (2008). https://doi.org/10.1007/s00221-008-1537-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-008-1537-z