Abstract
Background
Several studies indicate that organ failure is the leading cause of death in the postoperative phase after major surgery. An excessive inflammatory response followed by a dramatic depression of cell-mediated immunity after major surgery appears to be responsible for the increased susceptibility to subsequent sepsis. In view of this, most of the scientific and medical research has been directed towards measuring the progression and interrelationship of mediators after major surgery. Furthermore, the effect of those mediators on cell-mediated immune responses has been studied.
Objective
This article focuses on the effect of surgical injury and blood loss on cell-mediated immune responses in experimental studies utilizing models of trauma and hemorrhagic shock. The findings from those experimental studies will also be correlated with data from surgical patients.
Results
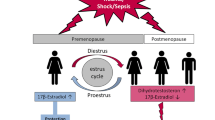
Recently, a gender-dimorphic immune and organ responsiveness in the susceptibility to and morbidity from shock, trauma, and sepsis has been found. Androgens have been shown to be responsible for the immunosuppression after trauma–hemorrhage in males. In contrast, female sex steroids exhibit immunoprotective properties after trauma and severe blood loss.
Conclusion
In view of these findings, clinically relevant therapeutic strategies have been developed using the testosterone receptor blocker flutamide and/or estrogen or agents with estrogenic effects, i.e., dehydroepiandrosterone, which might yield safe and useful therapeutic approaches for the treatment of immune depression in surgical patients.


Similar content being viewed by others
References
Carrico CJ, Meakins JL, Marshall JC (1986) Multiple organ failure syndrome. Arch Surg 121:196–208
Chaudry IH, Ayala A (1992) Immunological aspects of hemorrhage. Medical Intelligence Unit, R.G. Landes, Austin, TX, pp 1–132
Stephan RN, Ayala A, Chaudry IH (1993) Monocyte and lymphocyte responses following trauma. In: Schlag G, Redl H (eds) Pathophysiology of shock, sepsis and organ failure. Springer, Berlin Heidelberg New York, pp 131–144
Roumen RM, Hendriks T, van der Ven-Jongekrijg J, Nieuwenhuijzen GAP, Sauerwein RW, van der Meer JW, Goris RJA (1993) Cytokine patterns in patients after major surgery, hemorrhagic shock, and severe blunt trauma. Ann Surg 6:769–776
Faist E, Baue AE, Dittmer H (1983) Multiple organ failure in polytrauma patients. J Trauma 23:775–787
Levy EM, Alharbi SA, Grindlinger G, Black PH (1984) Changes in mitogen responsiveness lymphocyte subsets after traumatic injury: relation to development of sepsis. Clin Immunol Immunopathol 32:224–233
Keane RM, Birmingham W, Shatney CM, Winchurch RA, Munster AM (1983) Prediction of sepsis in the multitraumatic patient by assays of lymphocyte responsiveness. Surg Gynecol Obstet 156:163–167
Angele MK, Wichmann MW, Ayala A, Cioffi WG, Chaudry IH (1997) Testosterone receptor blockade after hemorrhage in males: restoration of the depressed immune functions and improved survival following subsequent sepsis. Arch Surg 132:1207–1214
Angele MK, Catania RA, Ayala A, Cioffi WG, Bland K, Chaudry IH (1998) Dehydroepiandrosterone (DHEA): an inexpensive steroid hormone which decreases the mortality from sepsis. Arch Surg 133:1281–1288
Knoferl MW, Angele MK, Diodato MD, Schwacha MG, Ayala A, Cioffi WG, Bland KI, Chaudry IH (2002) Female sex hormones regulate macrophage function after trauma–hemorrhage and prevent increased death rate from subsequent sepsis. Ann Surg 235:105–112
Menger MD, Vollmar B (2004) Surgical trauma: hyperinflammation vs immunosuppression? Langenbecks Arch Surg 389:475–484
Wang P, Ba ZF, Burkhardt J, Chaudry IH (1993) Trauma–hemorrhage and resuscitation in the mouse: effects on cardiac output and organ blood flow. Am J Physiol 264:H1166–H1173
Gutierrez G, Reines HD, Wulf-Gutierrez ME (2004) Clinical review: hemorrhagic shock. Crit Care 8:373–381
Moncada S, Higgs EA (1993) The l-arginine-nitric oxide pathway. N Engl J Med 329:2002–2012
Furchgott RF, Khan MT, Jothianandan D (1987) Evidence supporting the proposal that endothelium-derived relaxing factor is nitric oxide. Thromb Res 7:5
Weyrich AS, Ma X, Lefer AM (1992) The role of l-arginine in ameliorating reperfusion injury after myocardial ischemia in the cat. Circulation 86:279–288
McCall TB, Broughton-Smith NK, Palmer RM, Whittle BJ, Moncada S (1989) Synthesis of nitric oxide from l-arginine by neutrophils: release and interaction with superoxide anion. Biochem J 261:293–296
Yamauchi J, Vollmar B, Wolf B, Menger MD (1999) Role of TNF-alpha in local surgical trauma-induced microvascular dysfunction. Dig Surg 16:400–406
Ayala A, Lehman DL, Herdon CD, Chaudry IH (1994) Mechanism of enhanced susceptibility to sepsis following hemorrhage: interleukin (IL)-10 suppression of T-cell response is mediated by eicosanoid induced IL-4 release. Arch Surg 129:1172–1178
Nwariaku FE, McIntyre KL, Sikes PJ, Mileski WJ (1995) Alterations in alveolar macrophage tumor necrosis factor (TNF) response following trauma–hemorrhagic shock. Shock 4:200–203
Zellweger R, Ayala A, DeMaso CM, Chaudry IH (1995) Trauma–hemorrhage causes prolonged depression in cellular immunity. Shock 4:149–153
Wichmann MW, Zellweger R, Williams C, Ayala A, DeMaso CM, Chaudry IH (1996) Immune function is more compromised following closed bone fracture and hemorrhagic shock than hemorrhage alone. Arch Surg 131:995–1000
Haupt W, Riese J, Mehler C, Weber K, Zowe M, Hohenberger W (1998) Monocyte function before and after surgical trauma. Dig Surg 15:102–104
Faist E, Storck M, Hultner L, Redl H, Ertel W, Walz A, Schildberg FW (1992) Functional analysis of monocytes activity through synthesis patterns of pro-inflammatory cytokines and neopterin in patients in surgical intensive care. Surgery 112:562–572
Heidecke CD, Weighardt H, Hensler T, Bartels H, Holzmann B (2000) Immune paralysis of T-lymphocytes and monocytes in postoperative abdominal sepsis. Correlation of immune function with survival. Chirurg 71:159–165
Weighardt H, Heidecke CD, Emmanuilidis K, Maier S, Bartels H, Siewert JR, Holzmann B (2000) Sepsis after major visceral surgery is associated with sustained and interferon-gamma-resistant defects of monocyte cytokine production. Surgery 127:309–315
Trinchieri G (1993) Interleukin-12 and its role in the generation of TH1 cells. Immunol Today 14:335–338
O’Suilleabhain C, O’Sullivan ST, Kelly JL, Lederer J, Mannick JA, Rodrick ML (1996) Interleukin-12 treatment restores normal resistance to bacterial challenge after burn injury. Surgery 120:290–296
Weighardt H, Heidecke CD, Westerholt A, Emmanuilidis K, Maier S, Veit M, Gerauer K, Matevossian E, Ulm K, Siewert JR, Holzmann B (2002) Impaired monocyte IL-12 production before surgery as a predictive factor for the lethal outcome of postoperative sepsis. Ann Surg 235:560–567
Spolarics Z, Siddiqi M, Siegel JH, Garcia ZC, Stein DS, Denny T, Deitch EA (2003) Depressed interleukin-12-producing activity by monocytes correlates with adverse clinical course and a shift toward Th2-type lymphocyte pattern in severely injured male trauma patients. Crit Care Med 31:1722–1729
Ayala A, Perrin MM, Ertel W, Chaudry IH (1992) Differential effects of haemorrhage on Kupffer cells: decreased antigen presentation despite increased inflammatory cytokine (IL- 1, IL-6 and TNF) release. Cytokine 4:66–75
Ayala A, Perrin MM, Wang P, Ertel W, Chaudry IH (1991) Hemorrhage induces enhanced Kupffer cell cytotoxicity while decreasing peritoneal or splenic macrophage capacity: involvement of cell-associated TNF and reactive nitrogen. J Immunol 147:4147–4154
Pellegrini JD, Puyana JC, Lapchak PH, Kodys K, Miller-Graziano CL (1996) A membrane TNF-alpha/TNFR ratio correlates to MODS score and mortality. Shock 6:389–396
Ayala A, Ertel W, Chaudry IH (1996) Trauma-induced suppression of antigen presentation and expression of major histocompatibility class II antigen complex in leukocytes. Shock 5:79–90
Schinkel C, Sendtner R, Zimmer S, Faist E (1998) Functional analysis of monocyte subsets in surgical sepsis. J Trauma 44:743–748
Wakefield CH, Carey PD, Foulds S, Monson JR, Guillou PJ (1993) Changes in major histocompatibility complex class II expression in monocytes and T cells of patients developing infection after surgery. Br J Surg 80:205–209
MacLean LD, Meakins JL, Taguchi K, Duignan J, Dhillon KS, Gordon J (1975) Host resistance in sepsis and trauma. Ann Surg 182:207–211
Christou NV, Tellado JM (1993) The impact of preexisting disease conditions for host defense integrity in traumatized and critically ill patients. In: Faist E, Meakins J, Schildberg FW (eds) Host defense dysfunction in trauma, shock and sepsis. Springer, Berlin Heidelberg New York, pp 73–82
Stephan RN, Kupper TS, Geha AS, Baue AE, Chaudry IH (1987) Hemorrhage without tissue trauma produces immunosuppression and enhances susceptibility to sepsis. Arch Surg 122:62–68
O’Mahony JB, Palder SB, Wood JJ, McIrvine A, Rodrick ML, Demling RH, Mannick JA (1984) Depression of cellular immunity after multiple trauma in the absence of sepsis. J Trauma 24:869–875
Daniels JC, Sakai H, Cobb EK, Lewis SR, Larson DL, Ritzmann SE (1971) Evaluation of lymphocyte reactivity studies in patients with thermal burns. J Trauma 11:595–607
Abraham E, Chang Y-H (1985) The effects of hemorrhage on mitogen-induced lymphocyte proliferation. Circ Shock 15:141–149
Angele MK, Ayala A, Cioffi WG, Bland KI, Chaudry IH (1998) Testosterone: the culprit for producing splenocyte depression following trauma–hemorrhage. Am J Physiol 274:C1530–C1536
Hensler T, Hecker H, Heeg K, Heidecke CD, Bartels H, Barthlen W, Wagner H, Siewert JR, Holzmann B (1997) Distinct mechanisms of immunosuppression as a consequence of major surgery. Infect Immun 65:2283–2291
Heidecke CD, Hensler T, Weighardt H, Zantl N, Wagner H, Siewert JR, Holzmann B (1999) Selective defects of T lymphocyte function in patients with lethal intraabdominal infection. Am J Surg 178:288–292
Lyons A, Goebel A, Mannick JA, Lederer JA (1999) Protective effects of early interleukin 10 antagonism on injury-induced immune dysfunction. Arch Surg 134:1317–1323
Decker D, Schondorf M, Bidlingmaier F, Hirner A, von Ruecker AA (1996) Surgical stress induces a shift in the type-1/type-2 T-helper cell balance, suggesting down-regulation of cell-mediated and up-regulation of antibody-mediated immunity commensurate to the trauma. Surgery 119:316–325
Zedler S, Bone RC, Baue AE, von Donnersmarck GH, Faist E (1999) T-cell reactivity and its predictive role in immunosuppression after burns. Crit Care Med 27:66–72
Fuchs D, Gruber A, Wachter H, Faist E (1996) Activated cell-mediated immunity and immunodeficiency in trauma and sepsis. In: Faist E, Baue AE, Schildberg FW (eds) The immune consequences of trauma, shock and sepsis. Mechanisms and therapeutic approaches. Pabst Science, Lengerich, pp 235–239
Fuchs D, Malkovsky M, Reibnegger G, Werner ER, Forni G, Wachter H (1998) Endogenous release of interferon-gamma and diminished response of peripheral blood mononuclear cells to antigenic stimulation. Immunol Lett 23:103–108
Martin C, Boisson C, Haccoun M, Thomachot L, Mege JL (1997) Patterns of cytokine evolution (tumor necrosis factor-α and interleukin-6) after septic shock, hemorrhagic shock, and severe trauma. Crit Care Med 25:1813–1819
Xing Z, Gauldie J, Cox G, Baumann H, Jordana M, Lei XF, Achong MK (1998) IL-6 is an anti-inflammatory cytokine required for controlling local or systemic acute inflammatory responses. J Clin Invest 101:311–320
Tilg H, Trehu E, Atkins MB, Dinarello CA, Mier JW (1994) Interleukin-6 (IL-6) as an anti-inflammatory cytokine: induction of circulating IL-1 receptor antagonist and soluble tumor necrosis factor receptor p55. Blood 83:113–118
Sheeran P, Hall GM (1997) Cytokines in anaesthesia. Br J Anaesth 78:201–219
Strassmann G, Patil-Koota V, Finkelman F, Fong M, Kambayashi T (1994) Evidence for the involvement of interleukin 10 in the differential deactivation of murine peritoneal macrophages by prostaglandin E2. J Exp Med 180:2365–2370
Angele MK, Ayala A, Schwacha MG, Chaudry IH (2000) Effect of gender and sex on immune responses following shock. Shock 14:81–90
Wichmann MW, Zellweger R, DeMaso CM, Ayala A, Chaudry IH (1996) Enhanced immune responses in females as opposed to decreased responses in males following hemorrhagic shock. Cytokine 8:853–863
Angele MK, Nitsch SM, Knoferl MW, Ayala A, Angele P, Schildberg FW, Chaudry IH (2003) Gender specific p38 MAP kinase activation following trauma–hemorrhage: involvement of male and female sex steroids. Am J Physiol Endocrinol Metab 285:E189–E196
Diodato M, Knoferl MW, Angele MK, Schwacha MG, Bland KI, Chaudry IH (2001) Gender differences in the inflammatory response and survival following hemorrhage and subsequent sepsis. Cytokine 14:162–168
Angele MK, Ayala A, Monfils BA, Cioffi WG, Bland KI, Chaudry IH (1998) Testosterone and/or low estradiol: normally required but harmful immunologically for males after trauma–hemorrhage. J Trauma 44:78–85
Angele MK, Knoferl MW, Schwacha MG, Ayala A, Cioffi WG, Bland KI, Chaudry IH (1999) Sex steroids regulate pro- and anti-inflammatory cytokine release by macrophages after trauma–hemorrhage. Am J Physiol 277:C35–C42
Wichmann MW, Ayala A, Chaudry I (1997) Male sex steroids are responsible for depressing macrophage immune function after trauma–hemorrhage. Am J Physiol 273:C1335–C1340
Angele MK, Knoferl MW, Ayala A, Cioffi WG, Bland K, Chaudry IH (1998) Male and female sex steroids: do they produce deleterious or beneficial effects on immune responses following trauma–hemorrhage? Surg Forum 49:43–45
Mayr S, Wittmann F, Hernandez-Richter T, Angele P, Hatz RA, Faist E, Wichmann MW, Jauch KW, Angele MK (2003) Castration prevents suppression of MHC class II expression on macrophages following trauma–hemorrhage. Shock 19(Suppl):61
Knoferl MW, Diodato MD, Angele MK, Ayala A, Cioffi WG, Bland KI, Chaudry IH (2000) Do female sex steroids adversely or beneficially affect the depressed immune responses in males after trauma–hemorrhage? Arch Surg 135:426–433
Knoferl MW, Angele MK, Schwacha MG, Bland KI, Chaudry IH (2002) Preservation of splenic immune functions by female sex hormones after trauma–hemorrhage. Crit Care Med 30:888–893
Gannon CJ, Pasquale M, Tracy JK, McCarter RJ, Napolitano LM (2004) Male gender is associated with increased risk for postinjury pneumonia. Shock 21:410–414
Offner PJ, Moore EE, Biffl WL (1999) Male gender is a risk factor for major infections after surgery. Arch Surg 134:935–940
Oberholzer A, Keel M, Zellweger R, Steckholzer U, Trentz O, Ertel W (2000) Incidence of septic complications and multiple organ failure in severely injured patients is sex-specific. J Trauma 48:932–937
Wichmann MW, Angele MK, Ayala A, Cioffi WG, Chaudry I (1997) Flutamide: a novel agent for restoring the depressed cell-mediated immunity following soft-tissue trauma and hemorrhagic shock. Shock 8:1–7
Knoferl MW, Jarrar D, Angele MK, Ayala A, Schwacha MG, Bland KI, Chaudry IH (2001) 17 beta-Estradiol normalizes immune responses in ovariectomized females after trauma–hemorrhage. Am J Physiol Cell Physiol 281:C1131–C1138
Catania RA, Angele MK, Ayala A, Cioffi WG, Bland K, Chaudry IH (1998) Dehydroepiandrosterone (DHEA) restores immune function following trauma–hemorrhage by a direct effect on T-lymphocytes. Cytokine 11:443–450
Frantz MC, Prix NJ, Wichmann MW, van den Engel NK, Hernandez-Richter T, Faist E, Chaudry IH, Jauch KW, Angele MK (2005) Dehydroepiandrosterone (DHEA) restores the depressed PBMC function following major abdominal surgery via the estrogen receptors. Crit Care Med (in press)
Schneider C, von Aulock S, Zedler S, Schinkel C, Hartung T, Faist E (2004) Perioperative recombinant human granulocyte colony-stimulating factor (Filgrastim) treatment prevents immunoinflammatory dysfunction associated with major surgery. Ann Surg 239:75–81
Hartung T, von Aulock S, Schneider C, Faist E (2003) How to leverage an endogenous immune defense mechanism: the example of granulocyte colony-stimulating factor. Crit Care Med 31:S65–S75
Deitch EA (1998) Animal models of sepsis and shock: a review and lessons learned. Shock 9:1–11
Acknowledgements
These investigations were supported by a grant from the National Institutes of Health (R01 GM 37127) and a grant from the Deutsche Forschungsgesellschaft (DFG AN 357/1-1).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Angele, M.K., Chaudry, I.H. Surgical trauma and immunosuppression: pathophysiology and potential immunomodulatory approaches. Langenbecks Arch Surg 390, 333–341 (2005). https://doi.org/10.1007/s00423-005-0557-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-005-0557-4